食品安全一直是热点问题,尤其在全球人口众多的当今,确保食品安全变得比以往任何时候更加重要。传统的食品包装只注重对食品保护和保鲜等特点,而智能包装的推出为食品安全提供了重大保障,其不仅可延长食品的保质期,还可以利用先进的传感技术实时检测食品变质和污染的现象[1-2]。食品腐败会产生各种物质,如O2、CO2、挥发性盐基氮和H2S等,因此检测这些物质并能及时反馈是可视化监测食品腐败的关键[3-5]。
智能pH响应食品复合膜作为智能包装的一种,因其具有传统包装的保护和保鲜功能,同时兼具抗氧化及可实时监测食品腐败情况的功能,一直备受关注。近年来,人们开发了多种多样的智能pH响应食品复合膜,如SILVA-PEREIRA等[6]以壳聚糖和淀粉为成膜基材,以红甘蓝提取物为pH指示剂,制备了一种智能指示膜,该膜表现出良好的光学和形态学特性,可以根据pH值变化可视地指示鱼片变质情况。EZATI等[7]通过掺入茜素制备了壳聚糖基pH响应功能膜,并对其性能进行了活性和智能食品包装应用测试。DONG等[8]通过在琼脂/海藻酸钠聚合物基质中加入富含花青素的紫甘薯提取物和负载槲皮素的壳聚糖纳米颗粒,制备了一种抗氧化、抗菌和pH敏感的多功能薄膜,用于监测和保持虾的新鲜度。VISCUSI等[9]用大米淀粉/果胶/海藻酸盐为成膜基材,用姜黄和葡萄酒果渣的天然提取物为指示剂,开发了一种pH敏感性薄膜,该薄膜在食品变质过程中可以迅速发生颜色变化,是多用途智能食品包装的新型和可持续工具。这些膜有一个共同特点就是都可以实时监测食品腐败并及时反馈,这一特性是因为食品腐败会产生挥发性盐基氮,改变其pH环境,从而与智能pH响应食品复合膜的pH响应性材料发生显色变化,人们可以根据不同颜色变化而感知食品变质情况。所以智能pH响应食品复合膜能实时反映食品腐败情况的关键在于pH响应性材料,其分为人工合成材料(溴百里香酚蓝、甲酚红、甲基红等)和天然材料(花青素、姜黄素、茜素、紫草素、甜菜碱等),天然pH响应材料因其无毒、环保、低成本、易于制膜等特点被广泛用于制备智能pH响应食品复合膜。
智能pH响应食品包装膜主要由成膜基材和pH响应材料构成[10-11]。其中,成膜基材多种多样,但天然的pH响应材料却很有限。本文介绍了几种天然的pH响应材料(花青素、姜黄素、茜素、紫草素和甜菜碱)的来源、结构特性与提取工艺,阐述了智能pH响应食品复合膜的研究现状、制备方法以及应用进展,为该领域的进一步发展提供了全面参考。
1 天然pH响应材料的特性与提取工艺
1.1 常见天然pH响应材料
来源广泛、种类众多的天然色素多存在于植物的果实、叶片、根茎中,其具有的多样化功能和营养属性使其经常用于食品工业;此外,其还具有抗氧化、抗炎、抗癌、抗菌等多种生物活性,因此利用天然色素作为pH响应材料制备智能pH响应食品包装具有重大前景[11-12]。目前,已发现的天然pH响应材料主要包括花青素、姜黄素、茜素和紫草素等。其中,花青素因其在不同pH环境中显色明显且容易区分,被广泛用于制备智能pH响应食品包装,而其他天然pH响应材料也可以随环境pH的变化而发生不同显色变化,所以也可以当作智能pH响应食品包装的制备材料。
1.1.1 花青素
花青素广泛存在于自然界众多高等植物中,比如蓝莓、紫薯、葡萄、黑枸杞等,都是富含花青素的食物来源[13-15]。在这些植物中,花青素多存在于果实、花朵、叶子等部位,起着吸引昆虫传粉、抵御外界不良环境等作用。花青素属于类黄酮化合物,其基本结构是由2个苯环通过1个三碳的桥连结构连接而成(图1)[16]。不同种类的花青素其取代基有所差异,从而造就了多样的花青素类型,常见的有矢车菊素、飞燕草素、天竺葵素等类型,其结构的细微差别使其在理化性质等方面各有特点[17]。
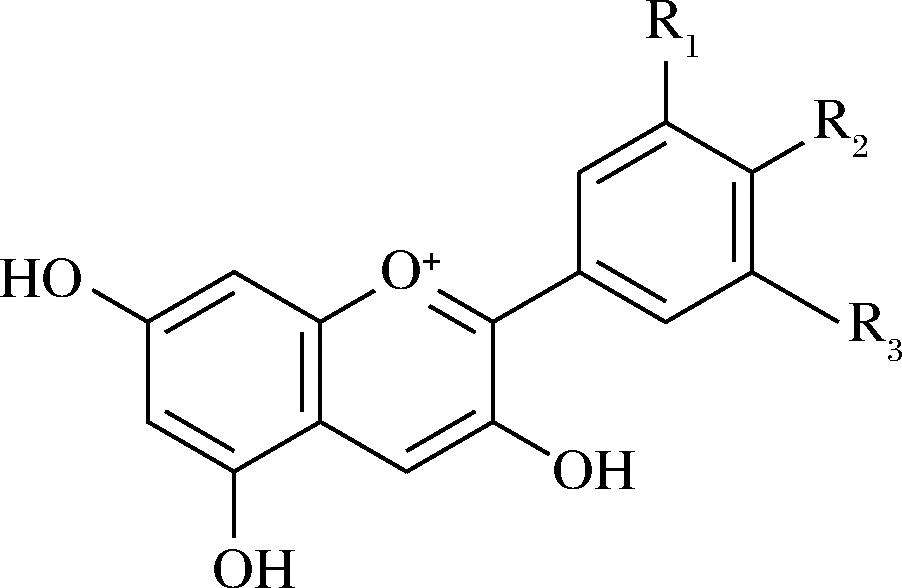
图1 花青素基本结构[16]
Fig.1 The basic structure of anthocyanin[16]
从颜色方面来看,花青素随所处溶液pH值不同而呈现出不同颜色,在酸性条件下(pH值<4.0)通常显红色,pH值在4.0~5.0时呈无色,中性时(pH 6.0~8.0)趋向蓝、紫色,碱性环境下(pH>8.0)则多呈现黄色,这也是一些花朵在不同酸碱土壤中呈现不同色彩的原因之一,图2是花青素在不同pH值下的结构图[18]。花青素易溶于水、甲醇、乙醇等极性溶剂,其对光、热、pH值等较为敏感,比如长时间光照、高温环境或者pH值剧烈变化时,容易发生降解,导致其含量和活性降低。此外,DEEPA等[19]研究发现花青素还具有多种生物学特性和健康益处,如抗氧化、抗菌、抗炎、抗糖尿病、抗肥胖、神经保护、抗癌等。
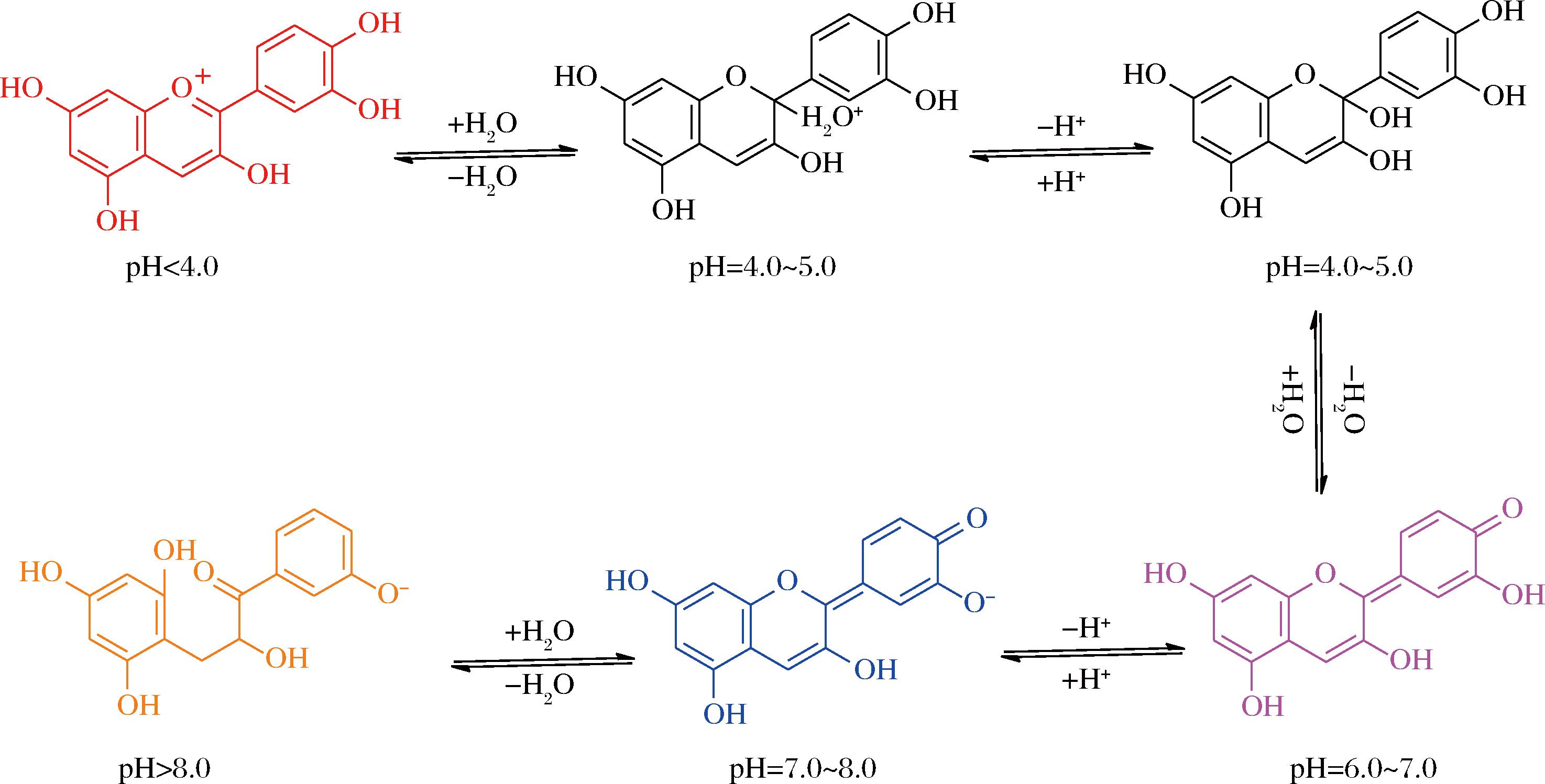
图2 不同pH值下的花青素结构图[18]
Fig.2 The structures of anthocyanins at different pH[18]
1.1.2 姜黄素
姜黄素是一种含有多种生物活性的天然β-二酮类的多酚化合物,其是从姜科植物(如姜黄、郁金和天南星科菖蒲等)根茎中提取出的。其分子结构包含2个苯环,中间通过1个含有不饱和双键的七碳链相连(图3),并且在苯环上还有羟基和甲氧基等取代基,这种特殊的结构赋予了姜黄素诸多化学性质[20-21]。
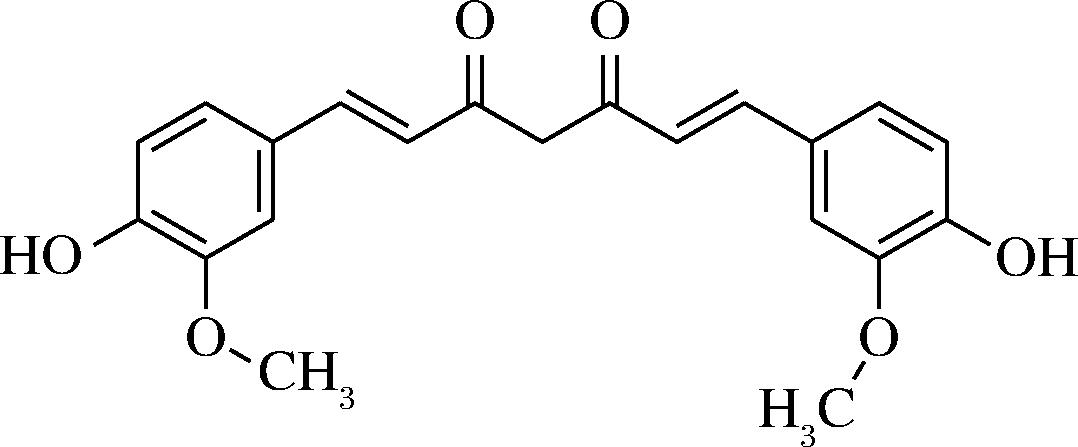
图3 姜黄素基本结构[21]
Fig.3 The basic structure of curcumin[21]
姜黄素是橙黄色结晶粉末,有特殊的气味,可溶于乙醇、丙酮等有机溶剂,在水中的溶解度较低。其在中性和酸性条件下(pH值<6)比较稳定,主要以酮式结构存在(图4-a),呈现亮黄色;在中性条件下(6
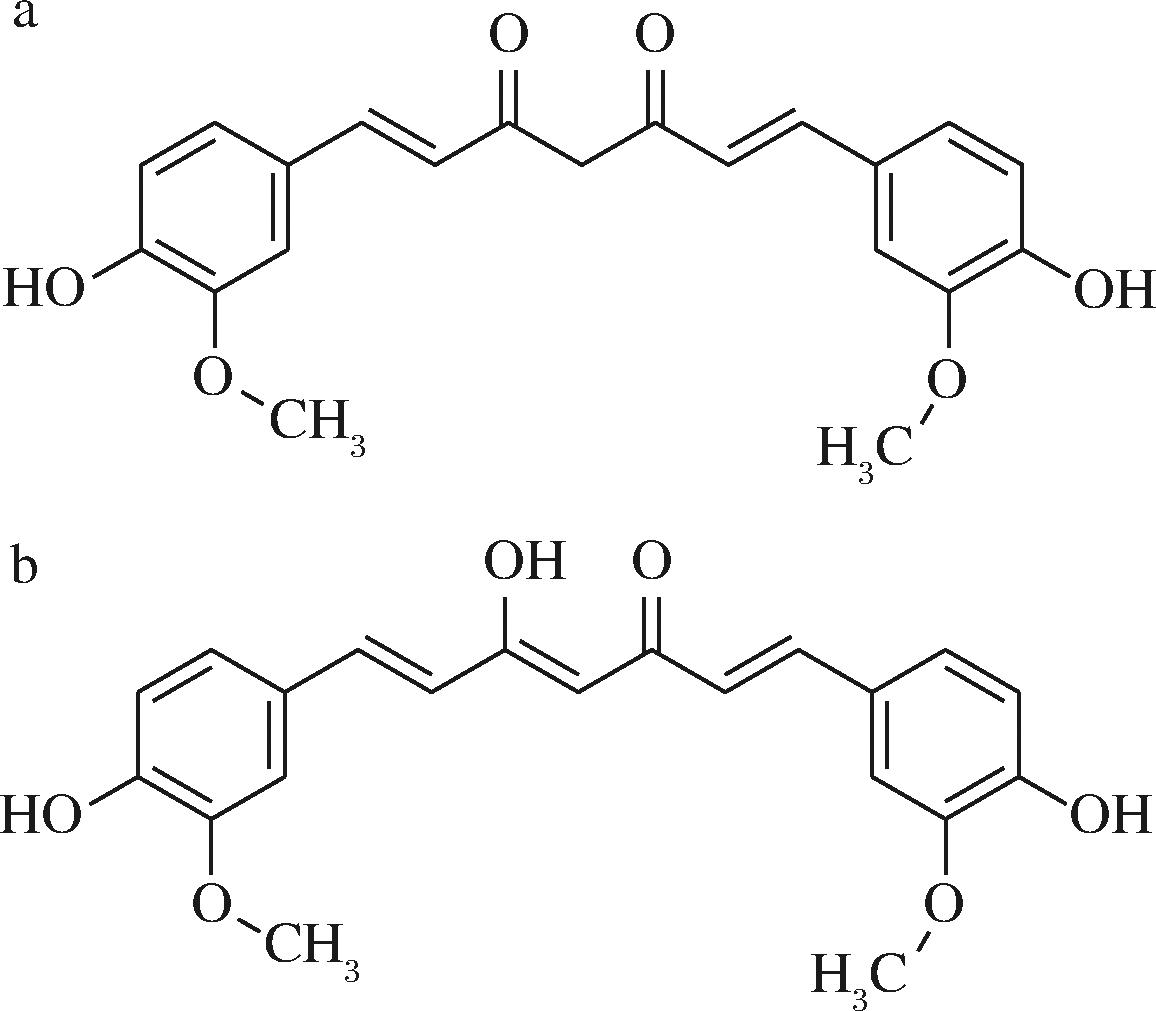
a-酮式结构;b-烯醇式结构
图4 姜黄素的酮、烯醇式结构图[22]
Fig.4 The ketone and enol structures of curcumin[22]
1.1.3 茜素
茜素(1,2-二羟基蒽醌)主要来源于天然茜草植物的根中,是天然染料的主要成分。其属于蒽醌类化合物,即由3个苯环稠合而成的有机化合物,并且在蒽醌的1位和2位上分别连接有羟基(图5)。该结构特点使茜素具有一定的化学活性,是其能发生多种化学反应的基础[25-26]。此外,茜素还有抗炎、抗病毒、抗菌和抗癌等作用,NGUYEN等[27]测试发现茜素对其自制的癌细胞系的抑制率超过90%。
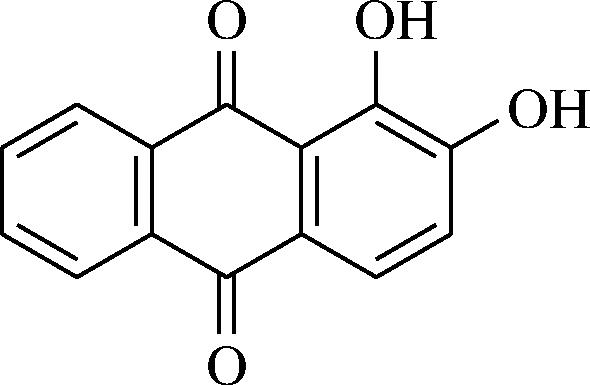
图5 茜素基本结构[25]
Fig.5 The basic structure of alizarin[25]
KHAN等[28]研究发现茜素在pH值≤6.0的溶液中呈黄色,在pH值为7.0~8.0的溶液中呈现紫红色,在pH值为9.0~12.0的溶液中呈现紫色,并且随着溶液pH值的增大,茜素的结构也发生相应的变化(图6)。
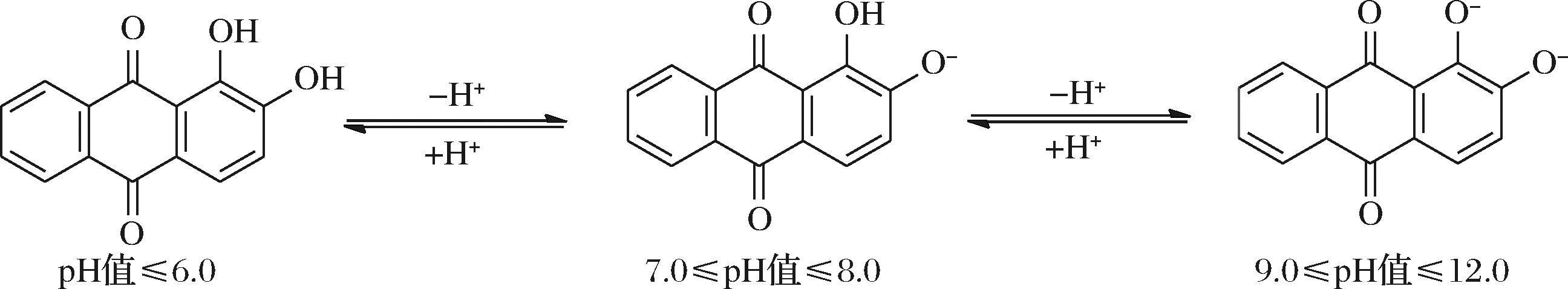
图6 不同pH值下的茜素结构图[28]
Fig.6 The structures of alizarins at different pH[28]
1.1.4 紫草素
紫草素是从紫草科植物根部中获得的。其是1,4-萘醌的衍生物,其化学结构主要是一个萘醌母核,在萘醌母核上连接有一些官能团(图7)。紫草素难溶于水,可溶于乙醇、氯仿等有机溶剂,在食品工业中,紫草素用来制备可生物降解的薄膜[29-32]。
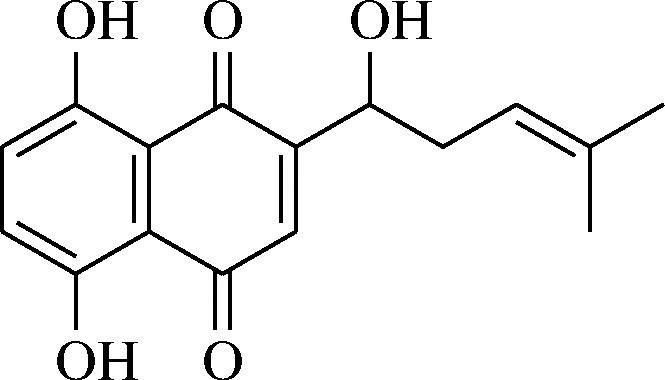
图7 紫草素基本结构[29]
Fig.7 The basic structure of shikonin[29]
紫草素在不同pH环境下会发生显著的颜色变化,在pH值为2~8时颜色变为鲜红色,pH值为8~10时由红紫色逐渐变为紫色,pH值>10时呈现蓝色,不同pH值下的结构如图8所示[33]。此外,因其主要成分具有抗炎、抗氧化、抗菌、免疫调节、抗癌、促进伤口愈合等药理活性,紫草素还常被用来开发药品,如ZHANG等[34]成功开发了pH响应型β-羟基异戊酰紫草素(SF@β-HIVS)纳米颗粒,验证了其治疗胰腺癌的潜力。
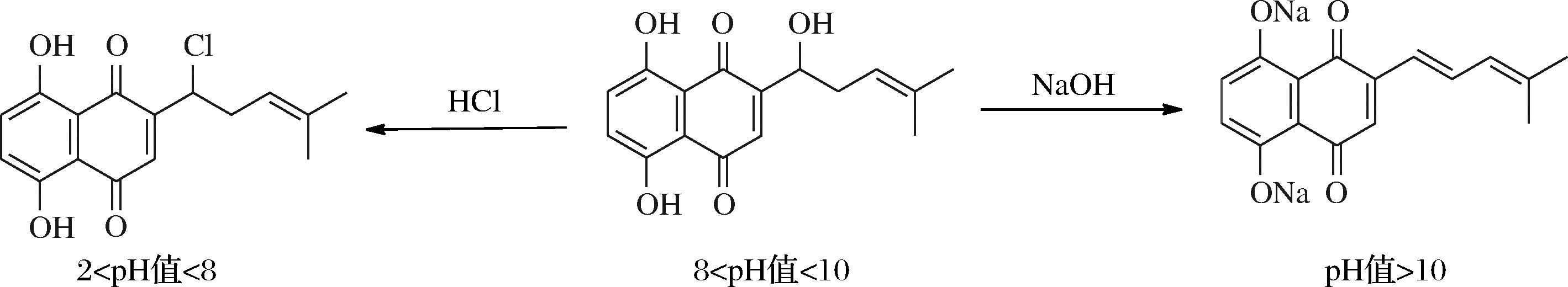
图8 不同pH值下的紫草素结构图[33]
Fig.8 Structure of shikonin at different pH[33]
1.1.5 甜菜碱
甜菜碱是一类含氮的水溶性色素,由酪氨酸衍生而来,存在于甜菜、菠菜、小麦麸皮、小麦胚芽和水生无脊椎动物中[35]。甜菜醛氨酸(图9-a)是存在于所有甜菜碱中的发色团,甜菜碱分为甜菜红素(图9-b)和甜菜黄素(图9-c),其结构划分取决于添加到甜菜酸上的残基,当甜菜酸与环-3,4-二羟基苯丙氨酸分子结合时形成甜菜红素,而与氨基酸(或胺)结合时则形成甜菜黄素[36]。甜菜碱是极性色素,颜色会从紫色到黄色/橙色再到红色变化。甜菜红素会在不同的pH值下显示不同的颜色,pH值<7时呈现红色/紫色,而pH值>8时则变为黄色/棕色[37]。此外,甜菜碱还对多种人类疾病具有有益作用,例如肥胖、糖尿病、癌症和阿尔茨海默病等[38]。
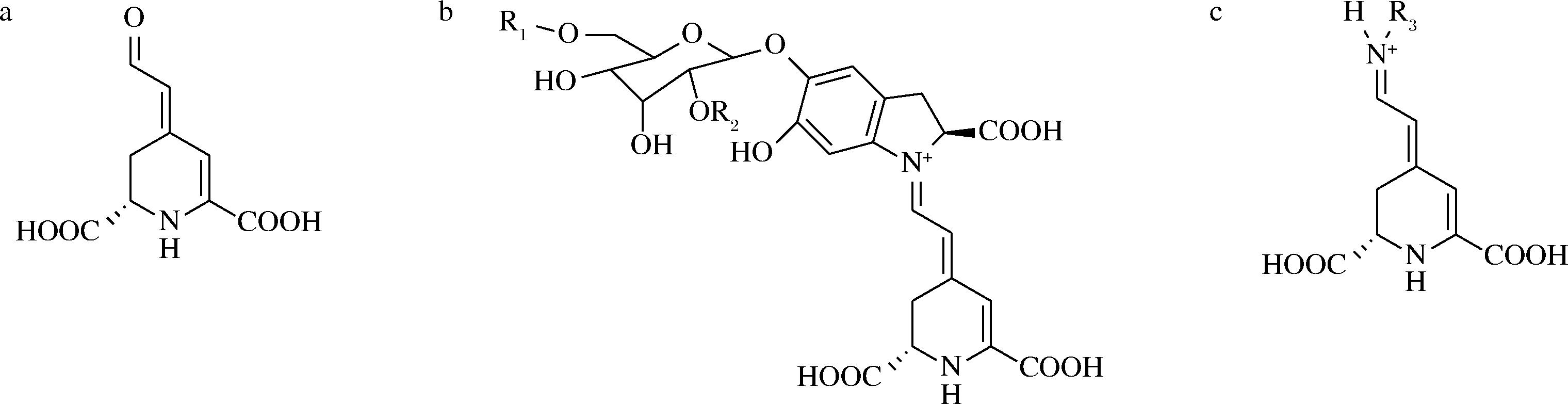
a-甜菜醛氨酸;b-甜菜红素;c-甜菜黄素
图9 甜菜醛氨酸与甜菜红素、甜菜黄素结构图[36]
Fig.9 The structures of betalamic acid, betacyanins and betaxanthins[36]
1.2 常见天然pH响应材料的提取方法
1.2.1 常见提取方法
天然色素存在于植物的各个部位(果实、叶片、根茎等),如何从植物中提取天然色素是一个难题。目前提取天然色素的方法主要有溶剂萃取法、酶解法、超临界流体萃取法和高压脉冲电场法等。其中溶剂萃取法又称固-液萃取法,在溶剂萃取法的基础上又发展出了超声波辅助法和微波辅助法[39]。天然色素中对pH响应性材料(花青素、姜黄素、茜素、紫草素、甜菜碱)的提取方法主要有固-液萃取法、超声波辅助法、微波辅助法3种。表1列出了天然pH响应材料不同提取方法的优缺点。
表1 天然pH响应材料不同提取方法的优缺点
Table 1 Advantages and disadvantages of different extraction methods of natural pH responsive materials
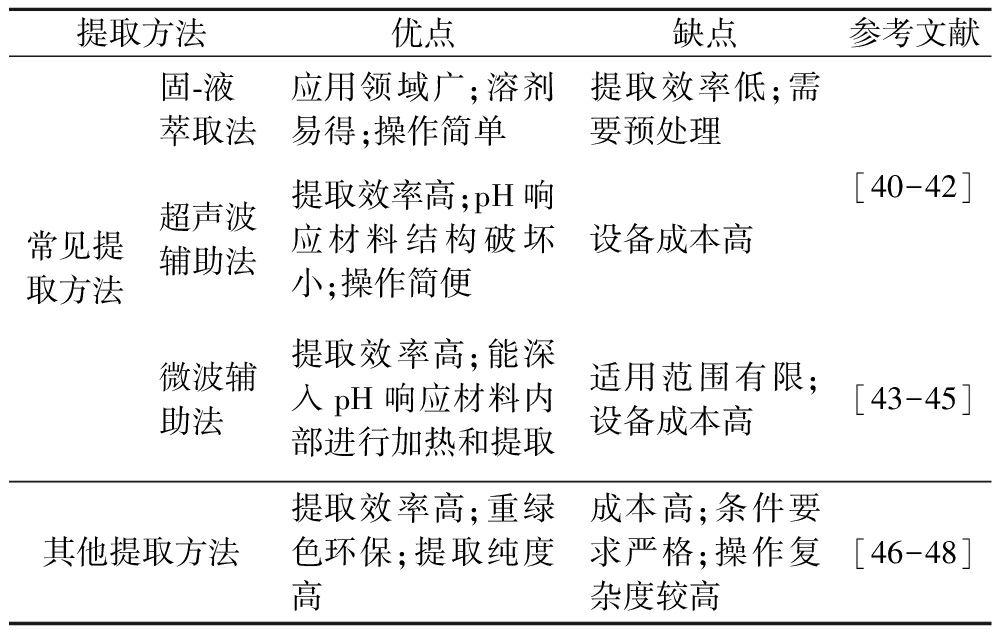
提取方法优点缺点参考文献常见提取方法固-液萃取法应用领域广;溶剂易得;操作简单提取效率低;需要预处理超声波辅助法提取效率高;pH响应材料结构破坏小;操作简便设备成本高微波辅助法提取效率高;能深入pH响应材料内部进行加热和提取适用范围有限;设备成本高[40-42][43-45]其他提取方法提取效率高;重绿色环保;提取纯度高成本高;条件要求严格;操作复杂度较高[46-48]
固-液萃取法是提取天然pH响应材料的传统手段,主要依据“相似相溶”原理,选择合适的溶剂对pH响应材料进行提取。大部分pH响应材料易溶于有机溶剂中,如LONGO等[40]用0.1 g/L HCl的甲醇溶液作为浸出溶剂萃取出了菝葜浆果中的花青素。DE SANTIS等[41]用甲醇萃取出了茜草根中的茜素,并对其染色棉和羊毛样品进行了染色评估。此外,LIU等[42]利用有机酸和糖形成的天然共晶溶剂在最佳的优化条件下提取天然色素姜黄素,其回收率高达93.2%。由此可见,固-液萃取法是有效的天然pH响应材料提取方法。
随着超声波技术和微波技术的发展,又出现了超声波辅助和微波辅助提取天然pH响应材料的方法。GHASSEMPOUR等[43]利用微波辅助和超声波辅助提取法从红皮葡萄的皮中提取出了花青素,2种方法的回收率高达90%和82%。KOYU等[44]利用微波辅助法提取黑天葵果实中的花青素,发现微波提取法比传统提取方法效果更好。除此之外,LEONARSKI等[45]比较了超声波辅助和微波辅助法从黑米糠中回收花青素的能力,结果证实超声波辅助法和微波辅助法都具有效率高、能耗小、耗时短的优点。
1.2.2 其他提取方法
除了上述主要的天然pH响应材料提取方法之外,酶解法、超临界流体萃取法和高压脉冲电场法等也可用于天然pH响应材料的提取。
酶解法主要是通过酶分解天然植物细胞壁的主要成分(如纤维素、半纤维素和果胶质等)使细胞内的天然pH响应材料溶于提取液中。SAHNE等[46]采用酶辅助离子液体的提取方法成功从姜黄中提取出了姜黄素,提取率比固-液萃取法(以丙酮作为常规有机提取溶剂)高近1倍。
而超临界流体萃取法主要通过调节超临界流体的压力和温度来改变其密度,使其提取出天然pH响应材料,与超临界流体萃取法不同,高压脉冲电场法主要通过将含有天然pH响应材料的植物置于高强度的电场中进行处理,改变或优化天然pH响应材料的性质,从而达到提取天然pH响应材料的目的。GACHOVSKA等[47]采用高压脉冲电场法从红甘蓝中提取花青素,发现通过脉冲电场处理,可将红甘蓝水中的总花青素提取量提高2.15倍,并证实这项技术可有效地从红甘蓝中提取花青素。此外,MIRZAZADEH等[48]采用了多种现代绿色萃取方法(如亚临界水萃取、脉冲电场萃取、超临界流体萃取和高静水压辅助萃取方法等),专门从红皮洋葱皮中提取和纯化花青素,结果发现高静水压辅助萃取和超临界流体萃取法提取的花青素浓度最高,这2种方法可增强花青素化合物保存。与传统提取方法相比这些提取方法更注重绿色环保、低能耗、高效率等。
2 智能pH响应食品复合膜的制备及其应用
2.1 智能pH响应食品复合膜的制备
智能pH响应食品复合膜主要采用一种或多种成膜基材(如聚乙烯醇、壳聚糖、海藻酸钠、明胶、淀粉等)与天然pH敏感性材料(花青素、姜黄素、茜素、紫草素、甜菜碱等)通过不同成膜方法(溶液浇铸法、静电纺丝法、溶胶-凝胶法、溶液流延法等)制备而成。到目前为止,常用的制备方法有溶液浇铸法和静电纺丝法。表2列出了不同成膜方法的优缺点。
表2 不同成膜方法的优缺点
Table 2 Advantages and disadvantages of different film-forming methods

成膜方法优点缺点参考文献溶液浇铸法操作简单;适用于实验室;制备的薄膜具有高拉伸强度和伸长率:大部分薄膜都可生物降解不适合大规模生产;生产效率较低;溶剂的挥发过程可能受到环境条件的影响[49-52]静电纺丝法工艺简单;制备的薄膜具有高疏水性、纤维直径小、孔隙率高、透气性好成本较高;制备过程不稳定;产率较低:不适合大规模生产;设备要求高[53-56] 溶胶-凝胶法工艺设备简单;适用于实验室工艺耗时;适用场景局限;薄膜有良好的吸光特性[57-58] 溶液流延法适用范围广;易于操作;可制备超薄膜环境控制要求高;设备要求高;成膜过程缓慢[59-60]
2.1.1 溶液浇铸法
溶液浇铸法是制备智能pH食品复合膜的传统方法。将成膜基材与pH敏感性材料溶解在适当的溶液里,通过搅拌等方式确保混合均匀,形成具有良好流动性的铸膜液,然后将膜液倒入特定模具中,干燥后形成智能pH食品复合膜[49]。MUSSO等[50]通过溶液浇铸法制备了明胶和花青素的智能可食用薄膜,此膜可用作食品的活性和智能包装,特别是用于易氧化或变质pH值随着变化的食品。WANG等[51]通过浇铸法制备了由山药黏液和淀粉组成的可食用薄膜,该膜无毒且可生物降解,在食品包装应用方面有巨大的潜力。ESFAHANI等[52]通过溶剂浇铸法制备了木薯淀粉和石榴皮粉的智能可食用薄膜,并用于羊肉新鲜度检测。用浇铸或涂布法制备智能pH响应食品复合膜简单方便,可广泛应用于生物聚合物,并且通过此方法制备的复合膜大部分都是可生物降解的,但也存在缺点,此方法不适合大规模加工食品复合膜。
2.1.2 静电纺丝法
静电纺丝法是制备智能pH响应食品复合膜的一种创新方法。该方法是将配制好的纺丝液(成膜基材与pH敏感材料的混合溶液)注射到静电纺丝机中,通过静电纺丝机制备成具有多孔结构的纤维膜[53]。KIM等[54]利用静电纺丝法制备了含姜黄素的聚乙烯醇/单宁酸纳米纤维膜,并用于虾腐败检测,结果发现此膜可以快速发生颜色变化,与传统方法制备的薄膜相比,复合纳米纤维膜由于其多孔和高表面积结构而表现出更快的颜色变化响应性,因此是一种高效和智能的指示膜。WANG等[55]利用静电纺丝法制备了花青素的玉米醇溶蛋白纤维膜,并用于畜牧和海产品的智能食品包装。该法制备的纳米纤维膜具有纤维直径小、孔隙率高、透气性好等优点,比传统铸造法制备的智能指标膜响应速度更快,能更高效、更快速地反映食品质量。然而,由于静电纺丝技术对设备的要求较高,操作技术不适合控制,与传统技术相比,成本增加,不能大规模推广,限制了静电纺丝技术在食品指标领域的发展[56]。
2.1.3 其他制备方法
除此上述方法外,溶胶-凝胶法、溶液流延法等也可以制备智能 pH 响应食品复合膜。
溶胶-凝胶法就是将成膜基材和天然pH响应材料溶于相应溶剂中,在溶液中将这些原料均匀混合,并进行水解、缩合反应,使其形成稳定的透明溶胶体系,溶胶经聚合,形成三维网络结构的凝胶,凝胶经过干燥就得到了复合膜[57]。TIRTASHI等[58]通过溶胶-凝胶法制备了纤维素/壳聚糖pH响应指示膜,通过此方法制备的复合膜可用作食品级生物材料,并且该膜可以通过颜色变化来判断牛奶是否变质。
溶液流延法是将成膜基材和天然pH响应材料溶解在相应溶剂中,然后通过流延的方式将其涂覆在培养皿或亚克力板上,最后通过干燥成膜[59]。AN等[60]通过溶液流延法制备了3种pH响应型智能显色指示剂膜,这3种纤维素基薄膜颜色都可以随着 pH 值的增加而发生变化从红紫色到浅蓝紫色,并使用大麻/紫草素指示膜用于虾新鲜度检测,有明显视觉变化。
2.2 智能pH响应食品复合膜的应用
使用天然pH响应材料和适合的可降解成膜基材,采用不同的成膜方法制成的智能食品复合膜,最终将用于监测新鲜食品。肉类食品变质特别容易改变其所处的pH环境,这会使智能pH响应食品复合膜发生变色,便于人们观察。此外,随着蔬菜、水果和乳制品的变质,智能pH响应食品复合膜也会发生明显的颜色变化。表3列出了不同类型智能pH响应食品复合膜在不同新鲜食品中的应用。
表3 不同类型智能pH食品复合膜在不同新鲜食品中的应用
Table 3 The application of different types of intelligent pH food composite films in various fresh foods
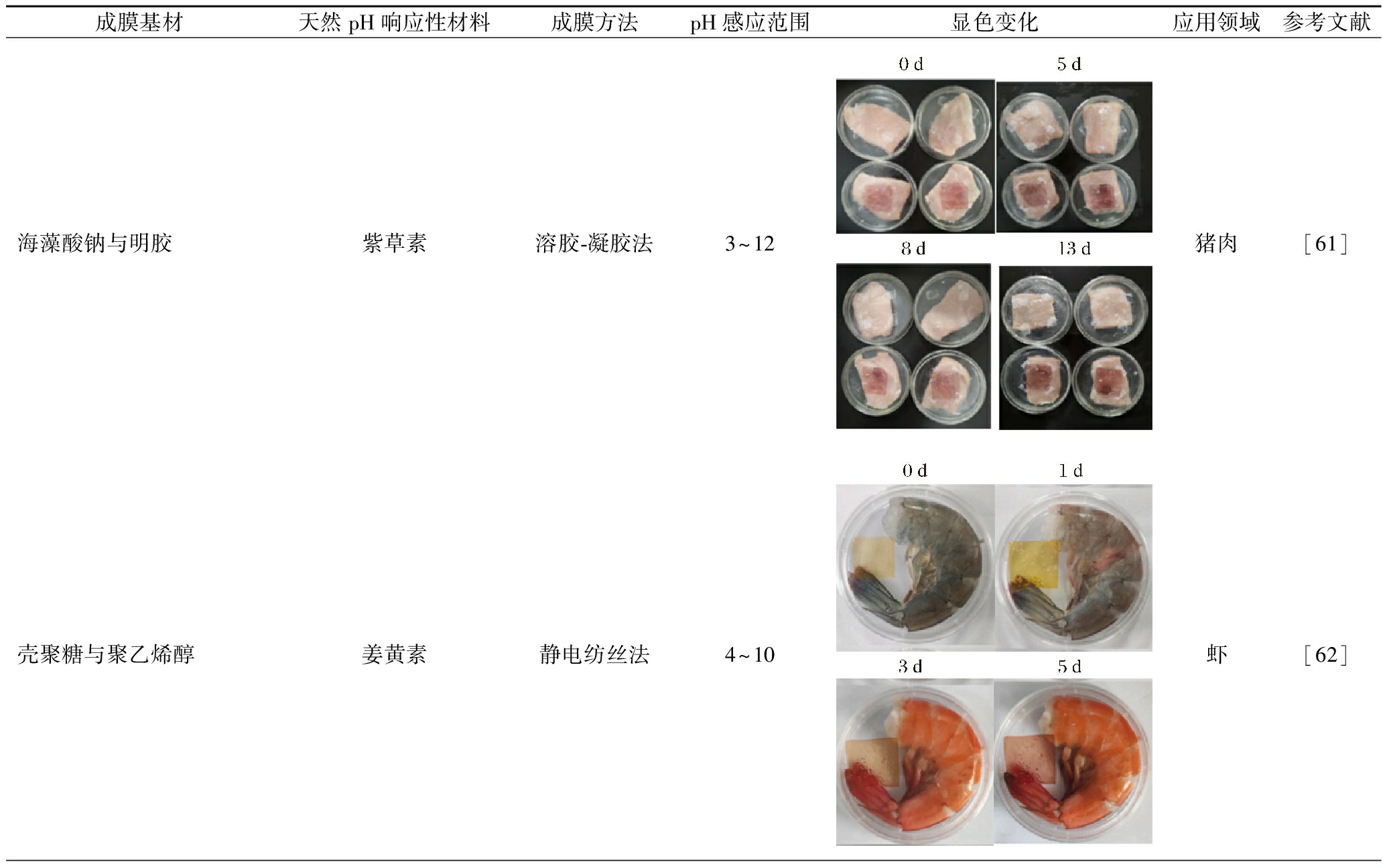
成膜基材天然pH响应性材料成膜方法pH感应范围显色变化应用领域参考文献海藻酸钠与明胶紫草素溶胶-凝胶法3~12猪肉[61]壳聚糖与聚乙烯醇姜黄素静电纺丝法4~10虾[62]
续表3

成膜基材天然pH响应性材料成膜方法pH感应范围显色变化应用领域参考文献海藻多糖与纤维素纳米晶茜素溶胶-凝胶法5~12三文鱼[63]壳聚糖与聚乙烯醇花青素溶液浇铸法2~10牛奶[64]聚乳酸与季铵化壳聚糖花青素静电纺丝法3~11蓝莓[65]聚乙烯醇与海藻酸钠-壳聚糖季铵盐花青素溶胶-凝胶法2~13牛奶[66]
2.2.1 肉类及水产品中的应用
随着人们生活水平的提高,人们对新鲜肉类的质量要求越来越高。肉类和水产品中含有丰富的脂肪、蛋白质和水分,特别适合微生物的生长和繁殖,导致变质。近年来,国内外学者对智能pH响应型食品包装膜在肉类及水产品的应用进行了大量的研究,发现随着肉类及水产品食物变质的过程,智能pH响应型食品包装膜可以实时发生颜色变化反映变质过程[67-68]。JIANG等[69]发现一些挥发性化合物,例如氨、三甲胺可用作肌肉食品质量下降的指标,而总挥发性氮含量与pH值密切相关。ZHOU等[70]制备了魔芋葡甘聚糖/茶花油和角叉菜胶/花青素/姜黄素的pH响应双层指示膜并用于鸡肉新鲜度监测,发现随着鸡肉的变质,挥发性氮含量也随之增加,pH值也相应增加,膜颜色发生变化,成功指示了鸡肉的变质过程。RADOOR等[71]通过溶剂浇铸法制备了加入荷花提取物的聚乙烯醇/海藻酸盐/大蒜薄膜,在虾储存过程中薄膜呈现出明显的从紫色到绿色的转变,表明虾已腐败,通过测量总挥发性氮也证实了这一结论,该薄膜有望成为实时食品监测应用的智能包装材料。
2.2.2 水果和蔬菜中的应用
水果和蔬菜是健康饮食中必需的食物,其既含有丰富的维生素,又含有大量的膳食纤维,可以促进胃肠道蠕动,促进消化。然而,蔬菜和水果也很容易变质,水果和蔬菜的腐败会产生多种代谢物,如肌酸、乙醇、挥发性含氮化合物等。水果和蔬菜的新鲜度可以根据这些物质的产量来评估。随着科学技术的不断进步,智能包装技术在果蔬保鲜中的应用显示出巨大的潜力[72]。XIA等[73]通过将聚乙烯醇与羧甲基纤维素混合并添加茜素和姜黄素开发了新型比色指示膜,用于常温和低温储存猕猴桃,发现该膜可以随着猕猴桃成熟腐败而实时发生颜色变化。ZHAN等[74]制备了基于含有红甘蓝花青素提取物的结冷胶新型比色膜,用于实时视觉检测蘑菇新鲜度,薄膜在蘑菇贮藏过程中的颜色变化在视觉上很容易区分,结果表明,该薄膜可以作为pH新鲜度智能标签来检测水果和蔬菜的新鲜度。
2.2.3 奶制品中的应用
奶制品是日常饮食中必需的,其含有丰富营养,但在储存过程中易受微生物及其自身酶的影响[75]。然而,由于乳制品本身的特殊性,其储存条件与其他食品不同,对智能包装指示剂比色法的研究较少,很多研究还处于试验阶段。KHODABAKHSH等[76]制备了含有红甘蓝提取物的壳聚糖/聚乙烯醇智能薄膜和含有红甜菜根皮提取物的壳聚糖/聚乙烯醇智能薄膜,2种智能膜在室温下可以实时发生颜色变化反映牛奶的腐败情况。
3 总结与展望
在全球人口众多的当今,确保食品安全变得比以往任何时候都重要。传统的食品包装只注重对食品保护和保鲜等特点,而智能包装的推出为食品安全提供了重大保障,其不仅可延长食品的保质期,也可以通过先进的传感技术来实时检测食品变质和污染的现象,智能pH响应食品复合膜作为智能包装的一种,因其不仅具有传统包装的保护和保鲜功能,还具有抗氧化及可实时监测食品腐败情况的功能,一直备受关注。本文介绍了几种天然的pH响应材料(花青素、姜黄素、茜素、紫草素和甜菜碱)的来源、结构特性与提取工艺,阐述了智能pH响应食品复合膜的研究现状、制备方法以及在肉类、水产品、奶制品和蔬菜水果中的应用进展,为该领域的进一步发展提供了全面参考。智能 pH响应食品复合膜作为一种新型的包装材料,具有体积小、灵敏度高、可实时直观监测的优点,适用于长期储存或长途运输时的食物质量监测,可在一定程度上减少食物浪费。然而,该类材料也面临一些挑战,如寻求更灵敏的天然pH响应材料、环保和廉价易得的新型成膜基材等;智能pH响应食品复合膜在储存食品时对食品的原有气味有没有影响;智能pH响应食品复合膜可能对使用此类膜产品的食品价格有所影响等。未来,不断优化智能pH响应食品复合膜的性能与材料可拓宽复合膜的应用领域,如在医疗卫生、环保、纺织工业等领域的应用。
[1] CHIU I, YE H X, AAYUSH K, et al.Intelligent food packaging for smart sensing of food safety[J].Advances in Food and Nutrition Research, 2024, 111:215-259.
[2] 童玲, 张剑.传统食品包装设计流程研究[J].食品与机械, 2016, 32(9):95-98.
TONG L, ZHANG J.The research of the packaging design process on traditional food[J].Food &Machinery, 2016, 32(9):95-98.
[3] WON K, JANG N Y, JEON J.A natural component-based oxygen indicator with in-pack activation for intelligent food packaging[J].Journal of Agricultural and Food Chemistry, 2016, 64(51):9675-9679.
[4] PULIGUNDLA P, JUNG J, KO S.Carbon dioxide sensors for intelligent food packaging applications[J].Food Control, 2012, 25(1):328-333.
[5] 周子萱, 杨迎澳, 任舒悦, 等.食品腐败相关气体检测研究进展[J].食品安全质量检测学报, 2023, 14(6):31-39.
ZHOU Z X, YANG Y A, REN S Y, et al.Recent advances in gas detection related to food spoilage[J].Journal of Food Safety and Quality, 2023, 14(6):31-39.
[6] SILVA-PEREIRA M C, TEIXEIRA J A, PEREIRA-J NIOR V A, et al.Chitosan/corn starch blend films with extract from Brassica oleraceae (red cabbage) as a visual indicator of fish deterioration[J].LWT, 2015, 61(1):258-262.
NIOR V A, et al.Chitosan/corn starch blend films with extract from Brassica oleraceae (red cabbage) as a visual indicator of fish deterioration[J].LWT, 2015, 61(1):258-262.
[7] EZATI P, RHIM J W.pH-responsive chitosan-based film incorporated with alizarin for intelligent packaging applications[J].Food Hydrocolloids, 2020, 102:105629.
[8] DONG S, ZHANG Y, LU D,et al.Multifunctional intelligent film integrated with purple sweet potato anthocyanin and quercetin-loaded chitosan nanoparticles for monitoring and maintaining freshness of shrimp[J].Food Packaging and Shelf Life, 2023, 35(000):11.
[9] VISCUSI G, LAMBERTI E, ANGIL F, et al.Smart pH-sensitive indicators based on rice starch/pectin/alginate loading Lambrusco pomace extract and curcumin to track the freshness of pink shrimps[J].International Journal of Biological Macromolecules, 2025, 291:139085.
F, et al.Smart pH-sensitive indicators based on rice starch/pectin/alginate loading Lambrusco pomace extract and curcumin to track the freshness of pink shrimps[J].International Journal of Biological Macromolecules, 2025, 291:139085.
[10] YAO Q B, HUANG F, LU Y H, et al.Polysaccharide-based food packaging and intelligent packaging applications:A comprehensive review[J].Trends in Food Science &Technology, 2024, 147:104390.
[11] WANG Y M, WU Y, CHEN Z X, et al.Intelligent food packaging materials:Principles, types, applications, and hydrophobization[J].Food Control, 2025, 171:111138.
[12] 邢金锋, 王稳航.天然色素的来源、分类、稳定化及其在可食包装中的应用[J].食品与发酵工业, 2021, 47(13):286-295.
XING J F, WANG W H.Source, classification, and stabilization of natural pigments and its application in edible packaging[J].Food and Fermentation Industries, 2021, 47(13):286-295.
[13] TAO W, DE LUCA L, MATEUS N, et al.A reliable, green and fast two-step solid-phase extraction methodology for the obtention of anthocyanin-rich extracts from different sources[J].Journal of Food Composition and Analysis, 2025, 140:107232.
[14] GAMAGE G C V, CHOO W S.Effect of hot water, ultrasound, microwave, and pectinase-assisted extraction of anthocyanins from black goji berry for food application[J].Heliyon, 2023, 9(3):e14426.
[15] HERN NDEZ-HERRERO J A, FRUTOS M J.Degradation kinetics of pigment, colour and stability of the antioxidant capacity in juice model systems from six anthocyanin sources[J].International Journal of Food Science &Technology, 2011, 46(12):2550-2557.
NDEZ-HERRERO J A, FRUTOS M J.Degradation kinetics of pigment, colour and stability of the antioxidant capacity in juice model systems from six anthocyanin sources[J].International Journal of Food Science &Technology, 2011, 46(12):2550-2557.
[16] XIAO Z C, KURTOVIC I, CHEN W F, et al.Kinetic analysis and starch digestion product composition reveal the subtle relationship between the anthocyanidin structure and inhibitory activity on pancreatic α-amylase[J].Journal of Agricultural and Food Chemistry, 2025, 73(7):4309-4319.
[17] KOSSYVAKI D, CONTARDI M, ATHANASSIOU A, et al.Colorimetric indicators based on anthocyanin polymer composites:A review[J].Polymers, 2022, 14(19):4129.
[18] DE OLIVEIRA FILHO J G, BRAGA A R C, DE OLIVEIRA B R, et al.The potential of anthocyanins in smart, active, and bioactive eco-friendly polymer-based films:A review[J].Food Research International, 2021, 142:110202.
[19] DEEPA P, HONG M J, SOWNDHARARAJAN K, et al.A review of the role of an anthocyanin, cyanidin-3-O-β-glucoside in obesity-related complications[J].Plants, 2023, 12(22):3889.
[20] TAHAY P, PARSA Z, ZAMANI P, et al.A structural and optical study of curcumin and curcumin analogs[J].Journal of the Iranian Chemical Society, 2022, 19(7):3177-3188.
[21] SHARMA R A, GESCHER A J, STEWARD W P.Curcumin:The story so far[J].European Journal of Cancer, 2005, 41(13):1955-1968.
[22] MART NEZ-GUERRA J, PALOMAR-PARDAVÉ M, ROMERO-ROMO M, et al.New insights on the chemical stability of curcumin in aqueous media at different pH:Influence of the experimental conditions[J].International Journal of Electrochemical Science, 2019, 14(6):5373-5385.
NEZ-GUERRA J, PALOMAR-PARDAVÉ M, ROMERO-ROMO M, et al.New insights on the chemical stability of curcumin in aqueous media at different pH:Influence of the experimental conditions[J].International Journal of Electrochemical Science, 2019, 14(6):5373-5385.
[23] LIU J R, WANG H L, WANG P F, et al.Films based on κ-carrageenan incorporated with curcumin for freshness monitoring[J].Food Hydrocolloids, 2018, 83:134-142.
[24] SHAHRAJABIAN M H, SUN W L.The golden spice for life:Turmeric with the pharmacological benefits of curcuminoids components, including curcumin, bisdemethoxycurcumin, and demethoxycurcumins[J].Current Organic Synthesis, 2024, 21(5):665-683.
[25] JIANG Y, NIU T C, WANG Z H, et al.Electrochemical polymerization of alizarin and the electrochemical properties of poly (alizarin)[J].Ionics, 2018, 24(5):1391-1397.
[26] DO K L, MUSHTAQ A, ZHAO F, et al.Alizarin old and new:Extraction techniques for coloration, advances in detection methods for historical textiles and novel applications as a functional dye[J].Coloration Technology, 2024, 140(1):5-29.
[27] NGUYEN T T H, PANDEY R P, PARAJULI P, et al.Microbial synthesis of non-natural anthraquinone glucosides displaying superior antiproliferative properties[J].Molecules, 2018, 23(9):2171.
[28] KHAN A, EZATI P, RHIM J W.Alizarin:Prospects and sustainability for food safety and quality monitoring applications[J].Colloids and Surfaces B:Biointerfaces, 2023, 223:113169.
[29] YADAV S, SHARMA A, NAYIK G A, et al.Review of shikonin and derivatives:Isolation, chemistry, biosynthesis, pharmacology and toxicology[J].Frontiers in Pharmacology, 2022, 13:905755.
[30] SONG Y P, DING Q T, HAO Y W, et al.Pharmacological effects of shikonin and its potential in skin repair:A review[J].Molecules, 2023, 28(24):7950.
[31] GAUTAM S, LAP
 K L, LAP
K L, LAP
 KOV
KOV B.Pharmacological significance of boraginaceae with special insights into shikonin and its potential in the food industry[J].Foods, 2024, 13(9):1350.
B.Pharmacological significance of boraginaceae with special insights into shikonin and its potential in the food industry[J].Foods, 2024, 13(9):1350.
[32] GUO Y M, ZHOU M M, MU Z Z, et al.Recent advances in shikonin for the treatment of immune-related diseases:Anti-inflammatory and immunomodulatory mechanisms[J].Biomedicine &Pharmacotherapy, 2023, 165:115138.
[33] ZOU Y C, SUN Y F, SHI W J, et al.Dual-functional shikonin-loaded quaternized chitosan/polycaprolactone nanofibrous film with pH-sensing for active and intelligent food packaging[J].Food Chemistry, 2023, 399:133962.
[34] ZHANG H, WANG Q, WANG S, et al.pH-sensitive silk fibroin nanoparticles encapsulating β-hydroxyisovaleryl shikonin for targeted pancreatic cancer therapy[J].Current Drug Delivery, 2025, https://doi.org/10.2174/0115672018342718241030070142
[35] DOBRIJEVI D, PASTOR K, NASTI
D, PASTOR K, NASTI N, et al.Betaine as a functional ingredient:Metabolism, health-promoting attributes, food sources, applications and analysis methods[J].Molecules, 2023, 28(12):4824.
N, et al.Betaine as a functional ingredient:Metabolism, health-promoting attributes, food sources, applications and analysis methods[J].Molecules, 2023, 28(12):4824.
[36] EYSHI S, GHAREAGHAJLOU N, AFSHAR MOGADDAM M R, et al.Red beet betalains extraction process:A comprehensive review of methods, applications, and physicochemical properties[J].Food Science &Nutrition, 2024, 12(11):8540-8558.
[37] KHAN M I, LIU J, SAINI R K, et al.Plant betalains-mixed active/intelligent films for meat freshness monitoring:A review of the fabrication parameters[J].Journal of Food Science and Technology, 2024, 61(7):1238-1251.
[38] ZHAO G F, HE F, WU C L, et al.Betaine in inflammation:Mechanistic aspects and applications[J].Frontiers in Immunology, 2018, 9:1070.
[39] 刘兴海, 邱诗波, 杜桂涛, 等.天然色素的提取、稳定性改进及其应用进展探究[J].数字印刷, 2022(1):1-25.
LIU X H, QIU S B, DU G T, et al.Research progress on extraction, stability improvement and application of natural pigments[J].Digital Printing, 2022(1):1-25.
[40] LONGO L, VASAPOLLO G.Extraction and identification of anthocyanins from Smilax aspera L.berries[J].Food Chemistry, 2006, 94(2):226-231.
[41] DE SANTIS D, MORESI M.Production of alizarin extracts from Rubia tinctorum and assessment of their dyeing properties[J].Industrial Crops and Products, 2007, 26(2):151-162.
[42] LIU Y H, LI J, FU R X, et al.Enhanced extraction of natural pigments from Curcuma longa L.using natural deep eutectic solvents[J].Industrial Crops and Products, 2019, 140:111620.
[43] GHASSEMPOUR A, HEYDARI R, TALEBPOUR Z, et al.Study of new extraction methods for separation of anthocyanins from red grape skins:Analysis by HPLC and LC-MS/MS[J].Journal of Liquid Chromatography &Related Technologies, 2008, 31(17):2686-2703.
[44] KOYU H, KAZAN A, DEMIR S, et al.Optimization of microwave assisted extraction of Morus nigra L.fruits maximizing tyrosinase inhibitory activity with isolation of bioactive constituents[J].Food Chemistry, 2018, 248:183-191.
[45] LEONARSKI E, KUASNEI M, DOS SANTOS E H, et al.Ultrasound and microwave-assisted extractions as green and efficient approaches to recover anthocyanin from black rice bran[J].Biomass Conversion and Biorefinery, 2025, 15(5):7251-7264.
[46] SAHNE F, MOHAMMADI M, NAJAFPOUR G D, et al.Enzyme-assisted ionic liquid extraction of bioactive compound from turmeric (Curcuma longa L.):Isolation, purification and analysis of curcumin[J].Industrial Crops and Products, 2017, 95:686-694.
[47] GACHOVSKA T, CASSADA D, SUBBIAH J, et al.Enhanced anthocyanin extraction from red cabbage using pulsed electric field processing[J].Journal of Food Science, 2010, 75(6):E323-E329.
[48] MIRZAZADEH N, BAGHERI H, MIRZAZADEH M, et al.Comparison of different green extraction methods used for the extraction of anthocyanin from red onion skin[J].Food Science &Nutrition, 2024, 12(10):7347-7357.
[49] ISLAM M H, HOSNA ARA M, KHAN M A, et al.Preparation of cellulose nanocrystals biofilm from coconut coir as an alternative source of food packaging material[J].ACS Omega, 2025, 10(9):8960-8970.
[50] MUSSO Y S, SALGADO P R, MAURI A N.Smart gelatin films prepared using red cabbage (Brassica oleracea L.) extracts as solvent[J].Food Hydrocolloids, 2019, 89:674-681.
[51] WANG R J, LI X J, LIU L, et al.Preparation and characterization of edible films composed of Dioscorea opposita Thunb.mucilage and starch[J].Polymer Testing, 2020, 90:106708.
[52] ESFAHANI A, MOHAMMADI NAFCHI A, BAGHAEI H, et al.Fabrication and characterization of a smart film based on cassava starch and pomegranate peel powder for monitoring lamb meat freshness[J].Food Science &Nutrition, 2022, 10(10):3293-3301.
[53] ZHANG J N, ZHANG J J, ZHOU L, et al.Two different fabrication method of high-hydrophobic Bi-layer indicators based on sodium alginate-polyvinyl alcohol/chitosan-zein incorporated with alizarin@ ZIF-8 for beef freshness visualization[J].International Journal of Biological Macromolecules, 2025, 307:141954.
[54] KIM J T, CHATHURANGA K, LEE J S, et al.Poly (vinyl alcohol)/tannic acid nanofibrous membrane containing curcumin as an intelligent indicator of food spoilage[J].Chemosphere, 2024, 369:143829.
[55] WANG J, ZHAO W P, QIN D L, et al.Incorporation of anthocyanin into zein nanofibrous films by electrospinning:Structural characterization, functional properties, and ammonia color-responsiveness[J].Food Chemistry:X, 2025, 25:102163.
[56] AMAN MOHAMMADI M, HOSSEINI S M, YOUSEFI M.Application of electrospinning technique in development of intelligent food packaging:A short review of recent trends[J].Food Science &Nutrition, 2020, 8(9):4656-4665.
[57] DRISSI S, EL KISSANI A, ABALI A, et al.A comparative study of sol-gel and electrodeposition methods used in the synthesis of Cu2FeSnS4 thin films[J].Optical Materials, 2025, 163:116972.
[58] TIRTASHI F E, MORADI M, TAJIK H, et al.Cellulose/chitosan pH-responsive indicator incorporated with carrot anthocyanins for intelligent food packaging[J].International Journal of Biological Macromolecules, 2019, 136:920-926.
[59] ZENG S L, LI L, WANG Q.Structure-property correlation of polyvinyl alcohol films fabricated by different processing methods[J].Polymer Testing, 2023, 126:108143.
[60] AN N, HU J W, DING Y, et al.Ionic liquid treated cellulose-based intelligent pH-responsive color indicator film, with excellent anti-ultraviolet function[J].Journal of Polymer Research, 2023, 30(9):343.
[61] ZHU J, ZHU Y, HE L C, et al.Designing of a new intelligent composite film based on sodium alginate/gelatin/shikonin complex for tracking meat freshness[J/OL].European Food Research and Technology, 2025.https://doi.org/10.1007/s00217-025-04700-6.
[62] HUANG X H, LI J M, HE J J, et al.Preparation of curcumin-loaded chitosan/polyvinyl alcohol intelligent active films for food packaging and freshness monitoring[J].International Journal of Biological Macromolecules, 2024, 276:133807.
[63] LI H, JIANG F, CHEN J, et al.Development of seaweed-derived polysaccharide/cellulose nanocrystal-based antifogging labels loaded with alizarin for monitoring aquatic products’ freshness[J].International Journal of Biological Macromolecules, 2023, 253:126640.
[64] RAJI M, EL FOUJJI L, MEKHZOUM M E M, et al.pH-indicative films based on chitosan-PVA/sepiolite and anthocyanin from red cabbage:application in milk packaging[J].Journal of Bionic Engineering, 2022, 19(3):837-851.
[65] XU M Y, FANG D L, SHI C, et al.Anthocyanin-loaded polylactic acid/quaternized chitosan electrospun nanofiber as an intelligent and active packaging film in blueberry preservation[J].Food Hydrocolloids, 2025, 158:110586.
[66] ZHANG Q, LIN G, WANG H N, et al.Development of smart packaging film incorporated with sodium alginate-chitosan quaternary ammonium salt nanocomplexes encapsulating anthocyanins for monitoring milk freshness[J].International Journal of Biological Macromolecules, 2024, 263:130336.
[67] 李波, 李文, 杨新, 等.基于紫薯花青素/茜素的猪肉新鲜度智能指示膜研究[J].食品与发酵工业, 2024, 50(16):160-168.
LI B, LI W, YANG X, et al.Development of intelligent indicator films based on purple sweet potato anthocyanin/alizarin for monitoring pork freshness[J].Food and Fermentation Industries, 2024, 50(16):160-168.
[68] FALLAH N, AARABI A, ZAKI DIZAJI H, et al.Development of a colorimetric intelligent film based on biodegradable polymers incorporated with Hibiscus sabdariffa anthocyanins for monitoring salmon fish spoilage[J].Food and Bioprocess Technology, 2025, 18(5):1-17.
[69] JIANG S, LIU Y.Gas sensors for volatile compounds analysis in muscle foods:A review[J].TrAC Trends in Analytical Chemistry, 2020, 126:115877.
[70] ZHOU X, YU X Z, XIE F, et al.pH-responsive double-layer indicator films based on konjac glucomannan/camellia oil and carrageenan/anthocyanin/curcumin for monitoring meat freshness[J].Food Hydrocolloids, 2021, 118:106695.
[71] RADOOR S, JAYAKUMAR A, KARAYIL J, et al.Nelumbo nucifera flower extract incorporated alginate/polyvinyl alcohol films as a sustainable pH indicator for active food packaging applications[J].International Journal of Biological Macromolecules, 2024, 273:133170.
[72] DU L Z, HUANG X W, LI Z H, et al.Application of smart packaging in fruit and vegetable preservation:A review[J].Foods, 2025, 14(3):447.
[73] XIA S, FANG X J, WU W J, et al.Preparation and characterization of novel natural pigment-indicating film:Application in kiwifruit freshness monitoring[J].Food Chemistry, 2025, 463(Part 4):141491.
[74] ZHAN S Q, YI F X, HOU F Y, et al.Development of pH-freshness smart label based on gellan gum film incorporated with red cabbage anthocyanins extract and its application in postharvest mushroom[J].Colloids and Surfaces B:Biointerfaces, 2024, 236:113830.
[75] POGHOSSIAN A, GEISSLER H, SCHÖNING M J.Rapid methods and sensors for milk quality monitoring and spoilage detection[J].Biosensors and Bioelectronics, 2019, 140:111272.
[76] KHODABAKHSH P, BAZARGANI-GILANI B.Comparison of pH and gas sensitivity of chitosan/polyvinyl alcohol smart film containing beet root peel and red cabbage indicators in milk spoilage monitoring[J/OL].Food Analytical Methods, 2025.https://doi.org/10.1007/s12161-025-02774-2.