“若作酒醴,尔唯曲糵”,好酒的源头是好曲[1]。酱香型大曲采用软质小麦粉碎后压制成曲块[2],并通过“堆曲”工艺(即稻草隔开曲块保温)实现高温培养(60~70 ℃)[3]。酱香型大曲在白酒酿造中兼具糖化、发酵与生香功能,其品质主要依赖于复杂微生物群落的协同作用[4]。
传统酱香型大曲的制备工艺具有深厚的历史渊源,其微生物群落通过自然接种形成。由于环境变量的不可控性及生物代谢网络的非线性特征,可能影响最终产品品质的一致性和稳定性。比如冬季低温干燥条件抑制群落演替速率,而夏季高温高湿环境促进优势菌群多样性富集,从而造成不同批次大曲质量差异性[5-6]。为此,许多学者通过分离酿造过程中的功能微生物、开发复合菌剂及优化原位培养条件,定向强化大曲的品质[7]。同时,学界正运用多组学联用技术系统解析核心功能菌株、菌群互作机理及代谢调控网络[8-9],从分子层面逐步掌握高温大曲发酵原理,为其生产优化提供理论支撑。当前,针对酱香型大曲强化的研究已聚焦于核心功能微生物多样性解析、分离与鉴定、定向强化策略构建等关键领域,但研究成果较为分散且缺乏系统性总结。
本文从酱香型大曲功能微生物的多样性组成出发,系统总结了可培养菌株的功能特征及基于非培养组学技术鉴定的功能菌株研究进展,深入分析了强化酱香型大曲的工艺特点及应用现状,并探讨了现有强化技术的局限性,以期为酱香型大曲的强化制备工艺优化以及产业应用提供科学参考。
1 酱香型大曲功能微生物的多样性组成
酱香型大曲制备参数(温度、湿度、发酵周期及贮藏时长等)对微生物群落组成及其动态演替具有显著调控作用。研究表明,高温高湿环境可诱导代谢通路的定向偏移,其中美拉德反应与酶促褐变现象与微生物代谢活动存在潜在协同关联[10]。此外,发酵仓内环境异质性(空间位置、温湿度及水分等)通过驱动微生物生态位分异,促使大曲形成黑、白、黄3种典型表型分化[11-12]。尽管制曲工艺(如机械与人工)[13]、母曲(如添加量与品质)[14]、制曲原料(如小麦与青稞)[15]及环境条件(如是否使用稻草)[16]等变量显著影响群落结构,但其核心功能菌群的组成仍呈现趋同特征。当前,针对酱香型大曲微生物群落结构的系统性研究主要基于传统培养方法、非培养组学测序技术、以及功能菌株的相互作用来展开。
1.1 基于培养法鉴定的功能菌株
酱香型大曲也被某些研究者称为“细菌曲”,原因在于,高温环境下大部分不耐高温的酵母菌和霉菌逐渐消亡,而细菌(尤其是耐热菌属)开始主导繁殖,形成独特微生物群落[17]。早期就有研究者采用传统可培养方法对酱香型大曲微生物群落的种属分布特征进行了系统研究。结果表明,其菌群组成以细菌为绝对优势类群(计数占比最高),其次为霉菌,而酵母菌与放线菌含量相对较低[18]。表1总结了酱香型大曲来源的功能微生物分离、鉴定情况以及功能特征。
表1 酱香型大曲功能微生物的分离、鉴定及功能特征
Table 1 Isolation, identification, and functional characteristics of functional microorganisms from sauce-flavor Daqu

由表1可知,酱香型大曲中功能微生物的筛选特点如下:细菌主要为产吡嗪(如四甲基吡嗪)或其前体物质为主的芽孢杆菌属(Bacillus),霉菌则为高产糖化酶、蛋白酶的曲霉属(Aspergillus)、根霉属(Rhizopus),酵母菌主要功能特征为产酯、产香及功能代谢物(如多元醇)能力,放线菌主要功能特征为耐高温、产酶及产香能力。
近年来,针对大曲功能微生物的研究已从单一提升酿造品质(如特征风味物质浓度、出酒率及酶活等方面)拓展至白酒安全领域。李超伟等[43]为了降低白酒发酵过程中有害副产物氨基甲酸乙酯(ethyl carbamate,EC)的含量,从大曲中筛选出一株B.amyloliquefaciens和一株W.anomalus,其EC降解率分别达到了85%和35%。曾玉雪等[44]利用高效液相色谱法检测生物胺,得到一株降解生物胺效果好的奥默柯达酵母(Kodamaea ohmeri)。SHEN等[45]采用元转录组学分析鉴定一株可产生腈酶的S.cerevisiae,将其接种于大曲中,最终氰化物含量降低了46.45%。
酱香型大曲及其酿造环境中所蕴含的微生物群落呈现高度的复杂多样性,通过传统培养手段仅能获取该环境中约0.1%~1.0%的微生物类群[46-47]。除传统培养方法外,培养组学通过优化培养条件与扩增子测序等技术结合,显著提升了微生物的可培养性。通过多种培养基优化策略联用基质辅助激光解吸电离飞行时间质谱与扩增子测序技术,LAGIER等[48]为人类肠道菌群增加了531种原核生物(含197个潜在物种)。在白酒领域,WANG等[49]基于不同pH梯度富集和筛选培养基与扩增子相结合,从酒醅中分离出38个细菌属,还有14个疑似新种。另有研究通过培养组学从浓香型窖泥中分离19个潜在新种[50]。朱丽萍等[51]通过培养基优化与扩增子分析,明确了清香型白酒各发酵阶段的主要霉菌种类。上述研究表明,未来可结合培养组学进一步挖掘酱香型大曲中微生物资源,为酿酒功能菌库的扩充提供技术支撑。
1.2 基于非培养法鉴定的功能菌株
近年来,酱香型大曲微生物群落解析已从传统可培养方法逐步向多组学技术分析拓展。通过高通量测序技术(包括扩增子测序、宏基因组学和宏转录组学)以及宏蛋白组学等多组学技术,再结合偏最小二乘回归、皮尔逊相关性等多元统计方法,对核心功能微生物进行功能预测和关联分析,揭示了大曲发酵过程中核心功能微生物的生态位分配及其代谢调控网络[6,52]。表2为基于多组学技术对不同酱香型大曲中功能菌株的潜在功能总结情况。
表2 基于多组学技术对酱香型大曲中功能菌株的研究总结
Table 2 Summary of functional strain studies in sauce-flavor Daqu based on multi-omics technologies

注:“—”表示无数据记录。
1.3 功能菌株的相互作用
功能菌株的筛选与鉴定为大曲强化奠定了基础。然而,微生物间的复杂互作同样在维持群落稳定性及驱动风味形成中扮演着关键角色[62]。需要注意的是,大曲制备环境的开放性和群落微生物组成的复杂性为深入解析这些互作机制带来了挑战。目前,相关研究方法主要分为非培养测序分析和菌株共培养实验。
非培养测序分析方法基于多组学测序数据(如宏基因组、宏转录组)结合统计学相关性分析,预测不同微生物类群间的潜在互作关系。例如,TONG等[63]对浓香型大曲关键发酵阶段的微生物群落演替、理化指标及咖啡酸产量进行相关性分析,发现葡萄球菌属(Staphylococcus)、克雷伯氏菌属(Klebsiella)、乳杆菌属(Lactobacillus)是参与咖啡酸代谢的关键功能类群;尤其在发酵前8 d菌株间互作增强,代谢活跃,表现出显著的咖啡酸生成潜力。DU等[64]对大曲发酵过程监测发现,部分耐热丝状真菌:蓝状菌属(Talaromyces)、Byssochlamys与部分细菌:Weissella、明串珠菌属(Leuconostoc)、链球菌属(Streptococcus)呈负相关,以及B.amyloliquefaciens和多数乳酸菌也呈负相关。WANG等[65]发现在清香型白酒发酵过程中,共生网络显示Lactobacillus和酵母菌属(Saccharomyces)在除了棒孢酵母属(Clavispora)外与其他优势微生物呈负相关,而其彼此之间显示出正相关关系。
菌株共培养实验策略依赖于可培养功能菌株离开原体系后进行的异位共培养,通过模拟原位条件来探究特定菌株间的互作机制。MENG等[66]将B.licheniformis与S.cerevisiae以不同接种比例于高粱提取液中共培养,发现S.cerevisiae抑制了B.licheniformis的生长,而后者却对S.cerevisiae的代谢具有促进作用;通过优化接种比例,共培养体系能有效提升乙醇及风味化合物的产量。程国富等[67]在固态发酵中发现,从酱香型大曲分离的B.cereus与S.cerevisiae按特定顺序(先接S.cerevisiae后接B.cereus)接种有利于共培养,并促进乙酸乙酯、乙酸、苯乙醇、2,3-二甲基-5-乙基吡嗪等风味物质的生成。另有研究表明,在30 ℃下,B.licheniformis能促进Z.bailii的乙醇转化率;但温度升至37 ℃时,前者会抑制后者生长并降低风味物质产量[68]。
此外,整合研究策略(即先通过测序分析预测关键互作,再利用共培养实验验证)也得到应用。TU等[69]在酱香型大曲发酵过程中,结合实时温度监测与绝对定量扩增子测序,并通过内聚指数(Cohesion index)和相关网络分析,揭示了大曲温度变化与片球菌属(Pediococcus)和Bacillus间互作存在显著关系。随后,该团队从大曲中分离出这两属的代表菌株,在模拟大曲环境发酵的异位(小麦碎培养基)上进行共培养,证实两者共存比单一菌属主导时能产生更多热量。ZHANG等[70]通过测序分析预测和共培养验证,在酱香型白酒发酵系统模型中确定了Pichia和丝状真菌之间的成对负相互作用,并推测Pichia的抗真菌活性可能是通过挥发性有机化合物(如2-苯乙醇)完成的。此外,该团队发现这种拮抗相互作用可以促进发酵生态位的建立。
综上所述,微生物间的相互作用是发酵过程中群落演替与代谢网络构建的重要基础。深入解析这些复杂的互作关系,不仅有助于深化对酱香型大曲复杂发酵过程的理解,更能为调控微生物群落结构、优化强化发酵工艺提供科学指导,从而定向生产具有独特风味和优良品质的酱香型大曲。
2 强化酱香型大曲的制备及其应用策略
2.1 强化酱香型大曲的制备
酱香型大曲采用高温制曲的制备工艺,将小麦转化为聚集了大量微生物群、多种酶类、丰富的挥发性风味及其前体物质于一体的酿酒中间产物,该制备过程涵盖人工或机械成块、高温发酵、贮藏等多个阶段[71]。强化酱香型大曲的制备即是利用内源性功能微生物强化原生微生态系统,先从酱香型高温大曲中通过不同条件分离出功能菌株,经活化扩培后制备成不同形态,并在不同制曲阶段进行添加(图1)。
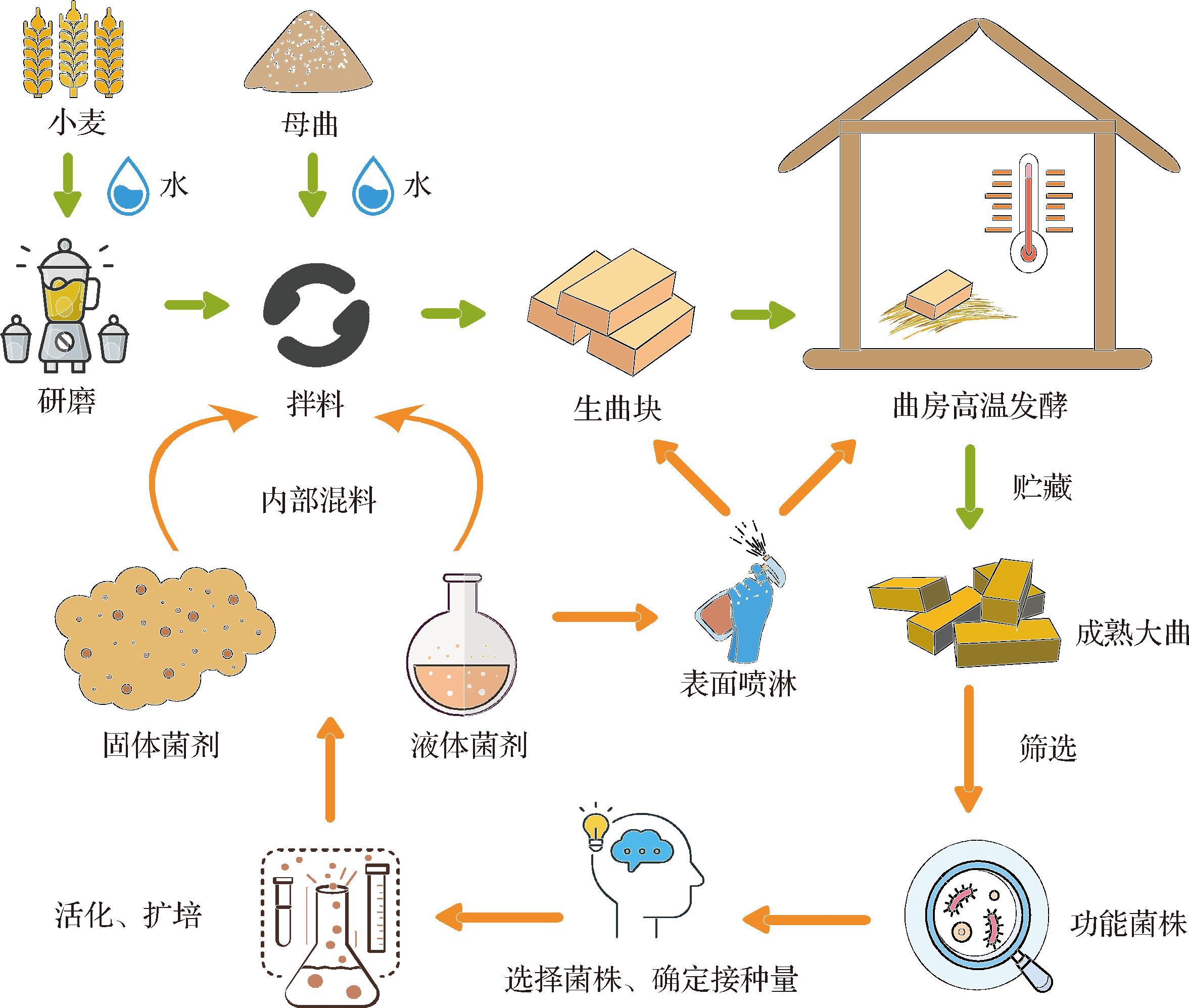
图1 制备强化酱香型大曲流程
Fig.1 The process of making fortified sauce-flavor Daqu
2.1.1 功能菌株制备形态
在强化大曲过程中,功能菌株常被制备成液体菌剂和固体菌剂2种形态进行添加。根据菌株特性,2种方式各有优点:液体菌剂可快速增殖菌体、菌种活性高、适合精准调控菌种比例;固体菌剂通过固态发酵或吸附工艺固定化微生物(如霉菌的孢子),防止发生生物污染。马鹏等[72]将红曲霉(Monascus purpureus)、A.oryzae、Rhizopus混合制备成固体米曲,替代部分大曲进行发酵,酒精度与挥发性物质含量均高于对照。ZHANG等[73]将B.velezensis制备成菌液喷洒在已经成型的陶融型大曲表面上发酵,生物扰动后大曲主要微生物群落发生变化,促进了淀粉、乙醇、脂肪酸和吡嗪的代谢,糖化和液化能力以及风味的含量显著增强,吡嗪含量增加了24.50%。卢延想[74]通过从高温大曲中分离出6株芽孢杆菌,将其分别制备成液体菌剂和固体菌剂,发现液态菌剂强化大曲产吡嗪效果普遍优于固态菌剂强化大曲。
2.1.2 功能菌株强化曲位
以往研究常采用均质强化方法,即直接将菌液[75]或者固体菌剂[76]接种在制曲物料搅拌阶段,然后压制成曲块。但均质强化法忽略了大曲不同层(如曲心、曲皮)中微生物的作用和组成。莫祯妮等[77]利用高通量测序解析酱香型大曲内部空间异质性,发现曲皮以链霉菌属(Streptomyces)、鞘氨醇单胞菌属(Sphingomonas)、Pediococcus为优势细菌属;曲心以火山渣芽孢杆菌属(Scopulibacilus)、Staphylococcus为优势细菌属。孙思佳等[78]通过表面喷洒和内部混料2种方式,用S.fibuligera来强化高温大曲,结果显示表面“穿衣”强化方式制备得到的大曲在糖化力、液化力、发酵力、品质等级等方面均优于内部混料大曲。因此,菌种强化曲位(即添加方式)直接影响菌株强化效果和最终曲的品质。未来,可进一步细化不同功能菌株类群的最优强化曲位,开发更加精准、高效的菌种强化策略。
2.1.3 功能菌株强化阶段
大多数研究进行菌株强化时,常选择在生曲制备阶段加入[79],忽略了高温制曲过程的长周期以及微生物复杂多样的变化。传统酱香型大曲高温制曲过程可分为4个步骤:曲坯入房、第一次翻曲、第二次翻曲和出房。郭敏[80]基于高通量测序得出Bacillus、Lactobacillus、Saccharopolyspora为酱香大曲制曲第一次翻曲阶段的优势细菌属,慢生芽孢杆菌属(Lentibacillus)、海洋杆菌属(Oceanobacillus)为酱香大曲制曲第二次翻曲阶段的优势细菌属;Thermomyces为第一次翻曲阶段的优势真菌属,Aspergillus、Byssochlamys为第二次翻曲阶段的优势真菌属。关于分阶段强化,许玲[81]选择高产淀粉酶并可疏松大曲内部的S.fibuligera、高产阿魏酸酯酶并代谢产生绵雅风味的谢瓦散囊菌(Eurotium chevalieri)和高产吡嗪类的B.subtilis共3株功能菌,开发出一种不同阶段强化高温大曲的制作工艺。此工艺将E.chevalieri液体菌剂与母曲、小麦碎混合后压制成曲块,再对压制好的大曲表面喷洒S.fibuligera菌液,当发酵至第一次翻曲(9 d后)时,再将B.subtilis菌剂喷洒于曲块表面。最终的强化曲表面黄褐色占比90%左右,曲皮较薄,曲的酱香味突出,醇类物质、酸类物质和吡嗪类物质的含量明显增多。所以功能菌株强化阶段的选择还有待更多的研究去发现。
2.2 强化酱香型大曲的应用效果
在强化酱香型大曲的制备过程中,外源菌株通常与母曲一起添加到物料中。根据添加菌株的数量,可分为单菌、双菌和多菌强化。菌株添加形态多为液体菌剂,而霉菌通常被制备成固体菌剂。目前,研究主要利用Bacillus属菌株来提升大曲中酯类、醇类、吡嗪类等物质的含量,也有部分研究采用霉菌、酵母菌以及多菌混合强化的方式,以提高大曲的风味质量与酶活力。表3总结了强化酱香型大曲中功能微生物的添加方式及曲的强化效果。
表3 强化酱香型大曲中的功能微生物添加及大曲的变化
Table 3 Enhancing the addition of functional microorganisms in sauce-flavor Daqu and the changes in Daqu

功能菌株的强化改善了大曲的酶活性和风味物质组成。当强化大曲进一步应用于实际白酒生产时,可显著提高出酒率,同时提升酒体中吡嗪类物质及挥发性风味物质含量。例如,LIU等[88]将高产纤维素酶的B.subtilis菌液喷洒于成型曲块表面制备强化大曲,用于固态模拟发酵后白酒产量提高3.36%。张柱等[89]将佛罗里达红曲霉(Monascus floridanus)制备为固体菌剂并添加至高温大曲,经模拟发酵,基酒产率及糟醅挥发性风味物质含量均获改善。张壮等[90]利用高产蛋白酶且产吡嗪性能优异的3株芽孢杆菌强化大曲,应用到4轮次白酒生产,酒体中三甲基吡嗪、四甲基吡嗪及糠醛含量显著提升。刘宇等[91]通过3株芽孢杆菌与2株霉菌的优化复配强化酱香大曲,让4轮次酒体的酯类、醇类、吡嗪类物质含量增加,感官上呈现突出的酱香特征及典型风格。郭小龙等[76]将B.licheniformis、S.cerevisiae、宛氏拟青霉(Paecilomyces variotii)、R.pusillus和T.crustaceus分别制备为固态菌剂并按不同比例添加至高温大曲,模拟发酵结果显示,酒醅中四甲基吡嗪及其他挥发性风味物质含量显著提高。
3 强化酱香型大曲的局限性
当前针对酱香型大曲发酵过程中部分微生物组成及其代谢功能的动态规律已逐步明晰。但研究体系仍存在显著局限性:首先,宏基因组学的检测技术虽可实现菌群组成的全面解析,但是仍缺乏活菌实时监测能力,导致大曲发酵过程动态调控存在滞后性;其次,菌株互作机制研究多局限于两菌之间在发酵系统外的分析,而对复杂菌群(不少于3株)的协同或拮抗效应鲜有报道。更关键的是,强化功能菌株的种属特性差异、生态位适应性、接种量以及接种曲位均可影响强化效果,为强化大曲的应用带来了一定的局限性。
菌株选择存在不确定性。目前已分离的菌株资源偏少,并且根据菌株特性选择强化目标也可能会失败。比如LI等[92]用能高产己酸乙酯的H.burtonii强化大曲,强化大曲中己酸乙酯的含量低于传统大曲,并没有表现出较好的酯化能力。同时功能菌株无法适应原生态位,被其他微生物竞争性抑制。比如只嗜热的菌难以在制曲初期富集,不耐热的菌在制曲中期会被抢占生态位,最终导致功能菌株无法发挥效果。
菌株的接种量以及接种曲位存在不确定性。第一,增加某些微生物种类到一定数量并影响到了核心微生物群对代谢组的贡献。PAN等[93]接种Bacillus后提高了大曲对芽孢杆菌类的富集,但抑制了乳酸杆菌和P.pastoris等亚群微生物的生长,虽然增加了酶活力以及吡嗪含量,但也显著降低了某些醇类物质的含量,影响最终风味。第二,由于大曲为固态发酵,同批大曲块乃至每一块大曲的不同部位都存在差异,造成曲坯和曲心的微生物形成差距,从而产生接种曲位选择(内部还是表面)等问题。
大曲制备环境是开放性的,如接种扩散能力强的菌株(如曲霉、根霉)可能引发不可预测的生态后果。因此在选择功能菌株时除了考虑其菌株特性、接种量、接种曲位,还要防止对大曲以及酿造环境产生不利影响。
4 结语与展望
本文首先探讨了酱香型大曲由于传统开放式制曲工艺导致品质稳定性受限,促使功能菌株定向强化技术成为近年研究热点。随后对酱香型大曲功能菌株多样性进行总结,对强化大曲工艺及应用效果进行解析,并系统梳理了当前强化酱香型大曲的进展。
基于现有研究,酱香型大曲的后续强化研究可重点关注以下方向:1)构建合成微生物群落:通过构建可控的合成微生物群落,可实现食品质量的定向调控。应结合培养组学优化菌株分离条件,利用多组学技术(如宏基因组、代谢组)定向筛选功能菌株,持续扩充可培养菌种资源库,为合成群落的构建提供科学依据。LU等[94]将26株来源于中高温大曲的本土微生物接种于碎小麦,形成更清洁的大曲,与传统大曲相比,该大曲成功复现传统大曲以酯类、醇类、酸类及醛类为主的风味特征,并展现出优于行业标准的糖化、液化和酯化活性。DU等[95]基于6株高产关键风味物质的菌株构建合成微生物群落,通过数学建模预测与实验验证,精准模拟了白酒关键风味成分。2)使用算法预测接种量以及曲位:可通过尝试不同的机器学习以及算法(随机森林、神经网络)来预测接种量以及接种曲位,实现强化工艺的精准调控。有研究使用Kriging代理模型对液态混菌比例进行优化,通过模型计算选择3组混菌比例进行验证实验,最终该模型预测含量与验证实验真实值的相对偏差仅小于10%[96]。3)开发新的强化大曲:除了使用野生功能菌株强化大曲,还可以尝试其他新技术来开发新的强化大曲(如太空大曲[97]、γ射线辐照[98]),以拓展强化大曲的创新路径。此外,未来行业亟需制定强化大曲制备以及质量的相关标准,以推动强化酱香型大曲制备从“经验依赖”到“精准可控”的跨越式升级。
[1] 周恒刚. “若作酒醴,尔惟曲糵”试解[J].酿酒科技, 1999, 95(5):22-23.ZHOU H G.Discussion on “Making liquor and drink only by koji and rice malt”[J].Liquor-Making Science & Technology, 1999, 95(5):22-23.
[2] ZHU Y Q, YANG Y, LU Z C, et al.Effect of wheat hardness on flavor compounds and microorganisms in medium-high temperature Daqu[J].CyTA-Journal of Food, 2025, 23(1):2465586.
[3] 吴煜樟, 郭敏杰, 高宇.酱香型白酒大曲生产使用稻草的研究进展[J].食品科技, 2023,48(6):62-66.WU Y Z, GUO M J, GAO Y.Research progress of straw used in production of Jiang-flavour Baijiu Daqu[J].Food Science and Technology, 2023,48(6):62-66.
[4] ZUO Q C, HUANG Y G, MINGUO.Evaluation of bacterial diversity during fermentation process:A comparison between handmade and machine-made high-temperature Daqu of Maotai-flavor liquor[J].Annals of Microbiology, 2020,70(1):57.
[5] YANG L, FAN W L, XU Y.Effects of storage period and season on the microecological characteristics of Jiangxiangxing high-temperature Daqu[J].Food Research International, 2024,196:115034.
[6] WANG L M, CHENG Y X, HU X X, et al.Analysis of bacterial diversity and functional differences of Jiang-flavored Daqu produced in different seasons[J].Frontiers in Nutrition, 2023,9:1078132.
[7] ZHAO H, DU B H, ZHAO P J, et al.Analysis of ester-producing performance in high-yield ethyl hexanoate yeast and the effect on metabolites in bio- enhanced Daqu, the starter for Baijiu and other traditional fermented foods[J].Journal of Applied Microbiology, 2024, 135(4):lxae081.
[8] 王国峥, 陈笔, 卢建军, 等.基于宏基因组学测序技术分析3个轮次高温大曲微生物群落[J].食品科学, 2023, 44(16):220-227.WANG G Z, CHEN B, LU J J, et al.Analysis of microbial communities in three rounds of high-temperature Daqu using metagenomic technology[J].Food Science, 2023, 44(16):220-227.
[9] 王玉荣, 侯强川, 田龙新, 等.基于纯培养和超高深宏基因组测序技术分析茅台镇两企业高温大曲微生物多样性差异[J].食品科学, 2024,45(4):108-115.WANG Y R, HOU Q C, TIAN L X, et al.Pure culture and ultra-high deep metagenomic sequencing for analysis of the differences in microbial diversity of high temperature Daqu from two distilleries in Maotai town[J].Food Science, 2024, 45(4):108-115.
[10] LUO S, ZHANG Q L, YANG F, et al.Analysis of the formation of sauce-flavored Daqu using non-targeted metabolomics[J].Frontiers in Microbiology, 2022, 13:857966.
[11] GAN S-H, YANG F, SAHU S K, et al.Deciphering the composition and functional profile of the microbial communities in Chinese Moutai liquor starters[J].Frontiers in Microbiology, 2019,10:1540.
[12] 梁二宏, 李金洋, 李微微, 等.3种高温大曲功能特征与微生物群落结构差异性分析[J].食品科学, 2024, 45(21):166-175.LIANG E H, LI J Y, LI W W, et al.Differences in functional characteristics and microbial community structures of three types of high-temperature Daqu[J].Food Science, 2024, 45(21):166-175.
[13] SHI G L, FANG C, XING S, et al.Heterogenetic mechanism in high-temperature Daqu fermentation by traditional craft and mechanical craft:From microbial assembly patterns to metabolism phenotypes[J].Food Research International, 2024,187:114327.
[14] 邓皖玉, 许永明, 陈波, 等.制曲工艺关键控制点对冬季高温大曲质量的影响[J].中国酿造, 2023, 42(8):153-157.DENG W Y, XU Y M, CHEN B, et al.Effect of key control points of Daqu-making process on the quality of high-temperature Daqu in winter[J].China Brewing, 2023, 42(8):153-157.
[15] 赵驰, 苏伟, 母应春, 等.不同原料对酱香大曲微生物群落结构及多样性的影响[J].食品科学, 2022,43(10):133-141.ZHAO C, SU W, MU Y C, et al.Effects of different raw materials on microbial community structure and diversity in Jiang-flavor Daqu[J].Food Science, 2022, 43(10):133-141.
[16] GONG L J, QIN S, ZHENG X R, et al.Traceability between microbial community and environmental microbial community in Maotai-flavor Daqu[J].Food Chemistry:X, 2025, 27:102321.
[17] YANG J G, DOU X, MA Y Y.Diversity and dynamic succession of microorganisms during Daqu preparation for Luzhou- flavour liquor using second-generation sequencing technology[J].Journal of the Institute of Brewing, 2018, 124(4):498-507.
[18] 王忠彦, 寇运同, 门芸, 等.高温大曲微生物区系的初步研究[J].酿酒科技, 1995(3):66-67.WANG Z Y, KOU Y T, MEN Y, et al.Preliminary study on microbial flora of high temperature Daqu[J].Liquor-Making Science & Technology, 1995, (3):66-67.
[19] 陈梦圆, 李志军, 罗爱民.高温大曲中高产四甲基吡嗪菌株的筛选及鉴定[J].酿酒科技, 2018(8):24-29.CHEN M Y, LI Z J, LUO A M.Screening and identification of strains with high yield of tetramethylpyrazine from high-temperature Daqu[J].Liquor-Making Science & Technology, 2018(8):24-29.
[20] 王庆, 王超彦, 徐海林, 等.高温大曲中高产四甲基吡嗪芽孢杆菌的分离鉴定及发酵条件优化[J].中国酿造, 2022,41(10):125-129.WANG Q, WANG C Y, XU H L, et al.Isolation and identification of high-yield tetramethylpyrazine Bacillus from high-temperature Daqu and optimization of fermentation conditions[J].China Brewing, 2022,41(10):125-129.
[21] 张璋, 赵腾飞, 李红霞, 等.产淀粉酶芽孢杆菌的分离鉴定、高温驯化及酶学性质研究[J].中国酿造, 2023, 42(9):79-84.ZHANG Z, ZHAO T F, LI H X, et al.Isolation, identification, high temperature acclimation and enzymatic property of amylase-producing Bacillus[J].China Brewing, 2023, 42(9):79-84.
[22] 李红霞, 王雄, 王新叶, 等.酱香型大曲中产淀粉酶系菌株的筛选鉴定及产酶条件优化[J].中国酿造, 2024, 43(12):124-130. LI H X, WANG X, WANG X Y, et al.Screening and identification of amylases-producing strain in sauce-flavor Daqu and optimization of the enzyme-producing conditions[J].China Brewing, 2024, 43(12):124-130.
[23] 张春林, 蒲春, 白从广, 等.高温大曲中产酱香风味细菌的筛选及其产香特性研究[J].食品科技, 2022, 47(5):1-6.ZHANG C L, PU C, BAI C G, et al.Screening and identification of Moutai flavor producing bacteria from high-temperature Daqu and its aroma producing characteristics[J].Food Science and Technology, 2022, 47(5):1-6.
[24] 邱勇, 王颖, 张煨, 等.高产四甲基吡嗪的菌株筛选及其麸曲制作工艺优化[J].中国酿造, 2023, 42(4):112-117.QIU Y, WANG Y, ZHANG W, et al.Strain screening of high-yield tetramethylpyrazine and optimization of its Fuqu process technology[J].China Brewing, 2023, 42(4):112-117.
[25] 刘续, 陆震鸣, 张晓娟, 等.高温大曲中大曲慢生芽孢杆菌的分离鉴定与特性分析[J].微生物学通报, 2025, 52(5):2189-2202. LIU X, LU Z M, ZHANG X J, et al.Isolation and characterization of Lentibacillus daqui from high-temperature Daqu[J].Microbiology China, 2025, 52(5):2189-2202.
[26] 王成俊, 李玲珊, 范梅, 等.酱香型大曲中产4-乙基愈创木酚芽孢杆菌的筛选、鉴定及特性研究[J].酿酒科技, 2023(4):45-52. WANG C J, LI L S, FAN M, et al.Screening, identification and characterization of 4-ethylguaiacol producing Bacillus strains from Jiangxiang Daqu[J].Liquor-Making Science & Technology, 2023(4):45-52.
[27] 张璋, 赵腾飞, 李红霞, 等.产淀粉酶芽孢杆菌的筛选、耐酸性驯化及淀粉酶酶学性质分析[J].中国酿造, 2024, 43(8):74-79. ZHANG Z, ZHAO T F, LI H X, et al.Screening and acid resistance acclimation of amylase-producing Bacillus and amylase characteristic analysis[J].China Brewing, 2024, 43(8):74-79.
[28] 蒋泽元, 万义芳, 蒲驰中, 等.高温大曲中高产淀粉酶和蛋白酶细菌的分离、鉴定及生物学特性研究[J].中国酿造, 2024,43(2):60-63.JIANG Z Y, WAN Y F, PU C Z, et al.Isolation, identification and biological characteristics of high-yield amylase and protease bacteria from high-temperature Daqu[J].China Brewing, 2024, 43(2):60-63.
[29] 范恩帝, 黄魏, 谢丹, 等.酱香大曲中高产四甲基吡嗪微生物的选育及应用研究[J].酿酒科技, 2022(11):45-50;71.FAN E D, HUANG W, XIE D, et al.Breeding and application of microorganisms with high yield of tetramethylpyrazine in Jiangxiang Daqu[J].Liquor-Making Science & Technology, 2022(11):45-50;71.
[30] 王晓丹, 徐佳, 周鸿翔, 等.酱香型大曲中分离到的阿姆斯特丹散囊菌产酶产香特性[J].食品科学, 2016, 37(11):154-159.WANG X D, XU J, ZHOU H X, et al.Characterization of Eurotium cristatus isolated from Maotai-flavored Daqu, a traditional Chinese Maotai-flavored liquor fermentation starter, for its abilities to produce enzymes and aromas[J].Food Science, 2016, 37(11):154-159.
[31] 郑钰涵. 高温大曲嗜热真菌分离鉴定及其性能与应用研究[D].淮安:淮阴工学院, 2023.ZHENG Y H.Research on the performance of isolation and identification of thermophilic fungus of high-temperature Daqu and its application[D].Huaian:Huaiyin Institute of Technology, 2023.
[32] 魏丕伟, 王凌云, 罗惠波, 等.大曲中高产酸性蛋白酶霉菌的分离鉴定[J].食品与发酵科技, 2014, 50(4):1-4;21.WEI P W, WANG L Y, LUO H B, et al.Isolation and identification of acid-protease producing fungi from Daqu[J].Sichuan Food and Fermentation, 2014, 50(4):1-4;21.
[33] 鲁珍, 王红照, 李恩中, 等.高温大曲中霉菌的筛选及其挥发性产物分析[J].酿酒科技, 2019(7):50-55.LU Z, WANG H Z, LI E Z, et al.Screening of a mold strain from high-temperature Daqu &identification of its volatile fermentation products[J].Liquor-Making Science & Technology, 2019(7):50-55.
[34] 蒋思峡, 邱树毅, 邹江鹏, 等.传统酱香大曲与机械化酱香大曲中酵母菌的初步研究[J].中国酿造, 2017, 36(3):59-65.JIANG S X, QIU S Y, ZOU J P, et al.Preliminary studies on yeasts from sauce-flavor Daqu made by traditional and mechanical methods[J].China Brewing, 2017, 36(3):59-65.
[35] 谯露. 酒曲中耐高温产香酵母菌在烤烟上部叶的定殖研究[D].贵阳:贵州大学, 2024.QIAO L.Colonization of high-temperature resistant aromatic yeast in the upper leaves of fluecured tobacco[D].Guiyang:Guizhou University, 2024.
[36] 章钰浛, 班世栋, 赵皓静, 等.温度对产香酵母产挥发性风味物质的影响[J].食品与发酵工业, 2022, 48(5):58-67.ZHANG Y H, BAN S D, ZHAO H J, et al.Effect of temperature on the volatile compounds produced by aroma producing yeasts[J].Food and Fermentation Industries, 2022, 48(5):58-67.
[37] 罗小叶, 王晓丹, 邱树毅.酱香大曲中3株放线菌的分离筛选及挥发性成分分析[J].中国酿造, 2018, 37(8):62-67.LUO X Y, WANG X D, QIU S Y.Isolation, screening and aroma component analysis of 3 strains of actinomycetes from Moutai-flavor Daqu[J].China Brewing, 2018, 37(8):62-67.
[38] 王芙蓉, 李靖, 赵益梅, 等.酱香大曲中高温放线菌的筛选及其产蛋白酶条件优化[J].中国酿造, 2022, 41(8):132-136.WANG F R, LI J, ZHAO Y M, et al.Screening of high-temperature actinomycetes in sauce-flavor Daqu and optimization of protease-producing conditions[J].China Brewing, 2022, 41(8):132-136.
[39] 王晓丹, 罗小叶, 邱树毅.茅台大曲中一株嗜热放线菌的分离筛选及特性研究[J].中国酿造, 2018, 37(4):51-56.WANG X D, LUO X Y, QIU S Y.Isolation and screening of a thermophilic actinomycete from Moutai-flavor Daqu and its characterization[J].China Brewing, 2018, 37(4):51-56.
[40] 于华, 黄丹, 陈卓, 等.酱香型大曲中产蛋白酶放线菌的分离及产酶条件研究[J].中国酿造, 2017, 36(2):64-68.YU H, HUANG D, CHEN Z, et al.Isolation of protease-producing actinomycetes from sauce—flavor Daqu and its protease-producing conditions[J].China Brewing, 2017, 36(2):64-68.
[41] 李豆南, 黄魏, 王晓丹, 等.酱香型大曲中高温放线的筛选及风味成分分析[J].食品科学, 2018, 39(6):171-176.LI D N, HUANG W, WANG X D, et al.Identification and flavor profile of a Thermoactinomycetaceae strain separated from Moutai-flavor Daqu[J].Food Science, 2018, 39(6):171-176.
[42] 王润楠, 朱华, 王坤, 等.酱香型白酒酿造过程中高温放线菌的筛选及其特性[J].食品研究与开发, 2023, 44(8):191-200.WANG R N, ZHU H, WANG K, et al.Screening and characterization of thermophilic actinomycetes isolated from sauce-flavor Baijiu[J].Food Research and Development, 2023, 44(8):191-200.
[43] 李超伟, 侯晓艳, 张宿义, 等.白酒大曲中氨基甲酸乙酯降解菌株的筛选及共发酵培养条件的优化[J].食品科学, 2025, 46(5):151-160.LI C W, HOU X Y, ZHANG S Y, et al.Screening of ethyl carbamate-degrading strains from Baijiu Daqu and optimization of co-fermentation conditions[J].Food Science, 2025, 46(5):151-160.
[44] 曾玉雪, 罗惠波, 余东, 等.浓香型大曲中降解生物胺菌株的筛选及应用[J].食品与发酵工业, 2021, 47(8):145-151.ZENG Y X, LUO H B, YU D, et al.Screening and application of biogenic amines degrading strain derived from Luzhou-flavor Daqu[J].Food and Fermentation Industries, 2021, 47(8):145-151.
[45] SHEN T, WU Q, XU Y.Biodegradation of cyanide with Saccharomyces cerevisiae in Baijiu fermentation[J].Food Control, 2021, 127:108107.
[46] STALEY J T, KONOPKA A.Measurement of in situ activities of nonphotosynthetic microorganisms in aquatic and terrestrial habitats[J].Annual Review of Microbiology, 1985, 39:321-346.
[47] VILANOVA C, PORCAR M.Are multi-omics enough?[J].Nature Microbiology, 2016, 1(8):16101.
[48] LAGIER J C, DUBOURG G, MILLION M, et al. Culturing the human microbiota and culturomics[J]. Nature Reviews Microbiology, 2018, 16(9): 540-550.
[49] WANG J X, HAO S Y, REN Q.Analysis of bacterial diversity in fermented grains of Baijiu based on culturomics and amplicon sequencing[J].Fermentation, 2023, 9(3):260.
[50] XU J L, SUN L P, XING X, et al.Culturing bacteria from fermentation pit muds of Baijiu with culturomics and amplicon-based metagenomic approaches[J].?偆iFrontiers in Microbiology, 2020, 11:1223.
[51] 朱丽萍, 杨强, 江威, 等.清香型小曲白酒霉菌菌群解析与酶活特性研究[J].食品与发酵工业, 2022, 48(7):70-77.ZHU L P, YANG Q, JIANG W, et al.Mold communities and enzyme activity characteristics in light-flavor Xiaoqu Baijiu[J].Food and Fermentation Industries, 2022, 48(7):70-77.
[52] HOU Q C, WANG Y R, CAI W C, et al.Metagenomic and physicochemical analyses reveal microbial community and functional differences between three types of low-temperature Daqu[J].Food Research International, 2022, 156:111167.
[53] 柳习月, 朱琪, 杨帆, 等.蛋白组学揭示苯丙氨酸代谢对酱香大曲风味影响[J].食品科技, 2021, 46(8):1-6.LIU X Y, ZHU Q, YANG F, et al.Proteomics reveals the effect of phenylalanine metabolism on the flavor of Daqu[J].Food Science and Technology, 2021, 46(8):1-6.
[54] JIN Y, LI D Y, AI M, et al.Correlation between volatile profiles and microbial communities:A metabonomic approach to study Jiang-flavor liquor Daqu[J].Food Research International, 2019, 121:422-432.
[55] XING S, SHI G L, LU J, et al.The discrepancy in amino acids within high-temperature Daqu:A novel metabolic marker for the quality evaluation of Daqu[J].Food Chemistry, 2025, 470:142645.
[56] XIA Y, ZHU M, DU Y K, et al.Metaproteomics reveals protein composition of multiple saccharifying enzymes in nongxiangxing Daqu and jiangxiangxing Daqu under different thermophilic temperatures[J].International Journal of Food Science &Technology, 2022, 57(8):5102-5113.
[57] YANG L, FAN W L, XU Y.Chameleon-like microbes promote microecological differentiation of Daqu[J].Food Microbiology, 2023, 109:104144.
[58] ZHANG Y D, XU J G, DING F, et al.Multidimensional profiling indicates the shifts and functionality of wheat-origin microbiota during high-temperature Daqu incubation[J].Food Research International, 2022, 156:111191.
[59] ZHU C T, CHENG Y X, SHI Q L, et al.Metagenomic analyses reveal microbial communities and functional differences between Daqu from seven provinces[J].Food Research International, 2023, 172:113076.
[60] ZHU Q, CHEN L Q, PENG Z, et al.The differences in carbohydrate utilization ability between six rounds of Sauce-flavor Daqu[J].Food Research International, 2023, 163:112184.
[61] 陈绍依, 郎莹, 邱树毅, 等.茅台镇不同区域酱香大曲微生物群落结构及生产性能对比[J].食品科学, 2023, 44(14):134-143. CHEN S Y, LANG Y, QIU S Y, et al.Comparative studies on microbial community structure and production performance of Jiang-flavor Daqu in different areas of Maotai Town[J].Food Science, 2023, 44(14):134-143.
[62] SMID E J, LACROIX C.Microbe-microbe interactions in mixed culture food fermentations[J].Current Opinion in Biotechnology, 2013, 24(2):148-154.
[63] TONG W H, LI Y Y, YANG Y, et al.Dynamic analysis Caffeic acid production driven by the key physicochemical factor and microbial community succession in Baijiu Daqu:A multi-microorganism fermentation of solid-state fermentation system[J].LWT, 2023, 190:115542.
[64] DU H, WANG X S, ZHANG Y H, et al.Exploring the impacts of raw materials and environments on the microbiota in Chinese Daqu starter[J].International Journal of Food Microbiology, 2019, 297:32-40.
[65] WANG S L, WU Q, NIE Y, et al.Construction of synthetic microbiota for reproducible flavor compound metabolism in Chinese light-aroma-type liquor produced by solid-state fermentation[J].Applied and Environmental Microbiology, 2019, 85(10):e03090-18.
[66] MENG X, WU Q, WANG L, et al.Improving flavor metabolism of Saccharomyces cerevisiae by mixed culture with Bacillus licheniformis for Chinese Maotai-flavor liquor making[J].Journal of Industrial Microbiology &Biotechnology, 2015, 42(12):1601-1608.
[67] 程国富, 郑自强, 卫春会, 等.蜡样芽孢杆菌与酿酒酵母混菌发酵效果探究[J].酿酒科技, 2021(3):53-59.CHENG G F, ZHENG Z Q, WEI C H, et al.Mixed fermentation effect of Bacillus cereus frankland and Saccharomyces cerevisiae[J].Liquor-Making Science & Technology, 2021(3):53-59.
[68] 庄孝杰, 吴群, 徐岩.酱香型白酒酿造拜耳接合酵母生理代谢特征及其与地衣芽孢杆菌相互作用[J].微生物学通报, 2017, 44(2):251-262.ZHUANG X J, WU Q, XU Y.Physiological characteristics of Zygosaccharomyces bailii and its interaction with Bacillus licheniformis in Chinese Maotai-flavor liquor making[J].Microbiology China, 2017, 44(2):251-262.
[69] TU H B, ZHOU G Y, LU J J, et al.Cooperative interaction between Pediococcus and Bacillus communities as a key factor in the high-temperature Thermal differentiation of Daqu[J].Food Bioscience, 2024, 62:105457.
[70] ZHANG H X, DU H, XU Y.Volatile organic compound-mediated antifungal activity of Pichia spp.and its effect on the metabolic profiles of fermentation communities[J].Applied and Environmental Microbiology, 2021, 87(9):e02992-e02920.
[71] 沈怡方. 白酒生产技术全书[M].北京:中国轻工业出版社, 1998.SHEN Y F.Liquor Production Technology Book[M].Beijing:China Light Industry Press, 1998.
[72] 马鹏, 何霞, 王丽玲, 等.复合霉菌产酯化酶工艺优化及强化发酵白酒挥发性代谢物差异研究[J].食品安全质量检测学报, 2023,14(14):144-154.MA P, HE X, WANG L L, et al.Optimization of esterifying enzyme production by compound mould and difference of volatile metabolites in intensified fermentation of Baijiu[J].Journal of Food Safety and Quality, 2023, 14(14):144-154.
[73] ZHANG P P, LIU Y B, LI H D, et al.Bioturbation effect of high - Yield pyrazine strain on the microbial community and flavour metabolites of fortified Daqu[J].LWT, 2024, 208:116711.
[74] 卢延想. 高温大曲中产香微生物的筛选及应用研究[D].天津:天津科技大学, 2021.LU Y X.Study on screening and application of fragrant producing microorganisms in high temperature Daqu[D].Tianjin:Tianjin University of Science and Technology, 2021.
[75] 梁慧珍, 卢延想, 刘正, 等.高温大曲中高产吡嗪类物质芽孢杆菌的筛选与应用[J].中国酿造, 2022, 41(1):116-122.LIANG H Z, LU Y X, LIU Z, et al.Screening and application of high-yield pyrazines Bacillus from high temperature Daqu[J].China Brewing, 2022, 41(1):116-122.
[76] 郭小龙, 邓灿, 张明春, 等.人工合成菌群替代传统母曲对高温大曲质量的影响[J].华中农业大学学报, 2024, 43(4):239-248. GUO X L, DENG C, ZHANG M C, et al.Effects of synthetic microbial communities replacing traditional Muqu on quality of high-temperature Daqu[J].Journal of Huazhong Agricultural University, 2024, 43(4):239-248.
[77] 莫祯妮, 邱树毅, 曾祥勇, 等.基于Illumina Hiseq高通量测序分析酱香型白酒大曲曲皮和曲心的细菌群落差异[J].中国酿造, 2022, 41(2):48-52.MO Z N, QIU S Y, ZENG X Y, et al.Difference of bacterial community in different parts of high-temperature Daqu based on Illumina Hiseq high-throughput sequencing[J].China Brewing, 2022, 41(2):48-52.
[78] 孙思佳, 翟磊, 许玲, 等.扣囊复膜孢酵母CICC 33077在芝麻香型白酒高温大曲生产中的应用[J].酿酒科技, 2018(7):76-82.SUN S J, ZHAI L, XU L, et al.Application of Saccharomycopsis fibuligera CICC 33077 in the production of high-temperature Zhimaxiang Daqu[J].Liquor-Making Science & Technology, 2018(7):76-82.
[79] HE G Q, DONG Y, HUANG J, et al.Alteration of microbial community for improving flavor character of Daqu by inoculation with Bacillus velezensis and Bacillus subtilis[J].LWT, 2019, 111:1-8.
[80] 郭敏. 基于高通量测序对酱香大曲制曲微生态多样性的研究[D].贵阳:贵州大学, 2018.GUO M.Study on microecologi cal diversity of Maotai flavor Daqu based on high-throughput sequencing[D].Guiyang:Guizhou University, 2018.
[81] 许玲. 国井绵雅酱香型白酒工艺研究[D].济南:齐鲁工业大学, 2019.XU L.Study on technology of Guojing Mianya Maotai-flavor Baijiu[D].Jinan:Qilu University of Technology, 2019.
[82] 周习, 云岭, 邱声强, 等.产酱香芽孢杆菌在酱香大曲中的生产应用研究[J].酿酒科技, 2022(3):89-92;96.ZHOU X, YUN L, QIU S Q, et al.Application of Jiangxiang flavor-producing Bacillus strains in the production of Jiangxiang Daqu[J].Liquor-Making Science & Technology, 2022(3):89-92;96.
[83] LIU X, FU J J, SHEN H J, et al.Oceanobacillus in high-temperature Daqu:Taxonomic diversity, metabolic characteristics and biofortification effect[J].Process Biochemistry, 2025, 150:68-79.
[84] 何猛超, 邬子璇, 西玉玲, 等.通过外源添加芽孢杆菌提升北方地区高温大曲的品质[J].食品工业科技, 2024, 45(1):145-154. HE M C, WU Z X, XI Y L, et al.Enhancing the quality of high-temperature daqu in northern China by adding exogenous Bacillus spores[J].Science and Technology of Food Industry, 2024, 45(1):145-154.
[85] 何琪, 张亚东, 晏永, 等.菌种发酵液添加对酱香型高温大曲品质的影响研究[J].酿酒科技, 2025(3):65-70.HE Q, ZHANG Y D, YAN Y, et al.Effects of adding bacterial fermentation broth on the quality of high-temperature Jiangxiang Daqu[J].Liquor-Making Science & Technology, 2025(3):65-70.
[86] 邓剑清. 酱香型白酒强化高温大曲工艺优化及比较研究[D].福州:福建师范大学, 2017.DENG J Q.The process optimization of Maotai liquor fortified high temperature Daqu and its comparative research[D].Fuzhou:Fujian Normal University, 2017.
[87] 颜林春. 高温大曲的细菌菌群结构分析和酿酒功能菌的选育及强化大曲的研究[D].福州:福建师范大学, 2012.YAN L C.The analysis of high-temperature daqu microbes structure and wine function bacteria breeding and theresearch of fortified Daqu[D].Fuzhou:Fujian Normal University, 2012.
[88] LIU Y B, LI H D, LIU W X, et al.Bioturbation analysis of microbial communities and flavor metabolism in a high-yielding cellulase Bacillus subtilis biofortified Daqu[J].Food Chemistry:X, 2024, 22:101382.
[89] 张柱, 黄钧, 周荣清, 等.高温大曲中功能红曲霉菌株的筛选与功能浅析[J].中国酿造, 2024, 43(4):53-61.ZHANG Z, HUANG J, ZHOU R Q, et al.Screening and function analysis of functional Monascus strains in high-temperature Daqu[J].China Brewing, 2024, 43(4):53-61.
[90] 张壮, 张德中, 何猛超, 等.秦池龙琬酱酒高温制曲工艺研究[J].酿酒, 2023, 50(3):135-138.ZHANG Z, ZHANG D Z, HE M C, et al.Study on high temperature koji making process of Longwan Jiang-flavour Chinese spirits[J].Liquor Making, 2023, 50(3):135-138.
[91] 刘宇, 管桂坤, 万自然, 等.复合功能微生物在酱香型白酒生产中的应用研究[J].酿酒科技, 2019(12):98-104;118.LIU Y, GUAN G K, WAN Z R, et al.Application of functional microorganisms in the production of Jiangxiang Baijiu[J].Liquor-Making Science & Technology, 2019(12):98-104;118.
[92] LI W W, FAN G S, FU Z L, et al.Effects of fortification of Daqu with various yeasts on microbial community structure and flavor metabolism[J].Food Research International, 2020, 129:108837.
[93] PAN Q L, HUANG J, ZHANG S Y, et al.Synergistic effect of biotic and abiotic factors drives microbiota succession and assembly in medium-temperature Daqu[J].Journal of the Science of Food and Agriculture, 2023, 103(9):4392-4400.
[94] LU Y J, YAN Q, ZHANG K Z, et al.Generation of a synthetic autochthonous microbiota responsible for the essential flavors and brewing properties of Daqu[J].Food Research International, 2024, 197:115180.
[95] DU R B, JIANG J, QU G Y, et al.Directionally controlling flavor compound profile based on the structure of synthetic microbial community in Chinese liquor fermentation[J].Food Microbiology, 2023, 114:104305.
[96] 刘昕, 吴天祥, 赵群丽, 等.基于Kriging代理模型对产不饱和脂肪酸的酒曲微生物混菌比例优化[J].酿酒科技, 2016(9):23-27.LIU X, WU T X, ZHAO Q L, et al.Optimization of mixed ratio of microbial strains to produce unsaturated fatty acids based on Kriging model[J].Liquor-Making Science & Technology, 2016(9):23-27.
[97] MU Y, HUANG J, ZHOU R Q, et al.Comprehensive analysis for the bioturbation effect of space mutation and biofortification on strong-flavor Daqu by high-throughput sequencing, volatile analysis and metabolomics[J].Food Chemistry, 2023, 403:134440.
[98] ZENG Y, QIU Y, WANG Y, et al.Effects of γ-ray irradiation on insect pests, physicochemical properties, and microbial community of strong-flavor Daqu during maturation[J].Applied Radiation and Isotopes, 2025, 220:111780.